
Modified Mastering Chemistry with Pearson eText -- Standalone Access Card -- for Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134261430
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 10.7, Problem 31P
(a)
Interpretation Introduction
Interpretation:
The stereo isomeric product should be given when the
Concept introduction:
An alkene undergoes an oxidation reaction with Osmium tetra oxide and followed by hydrolysis to give a cis 1, 2
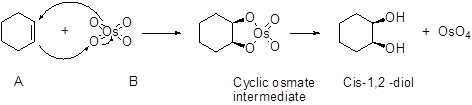
(b)
Interpretation Introduction
Interpretation:
The stereo isomeric product should be given when the alkene reaction with osmium tetraoxide followed by hydrogen peroxide hydrolysis.
Concept introduction:
An alkene undergoes an oxidation reaction with Osmium tetra oxide and followed by hydrolysis to give a cis 1, 2 diol for example,
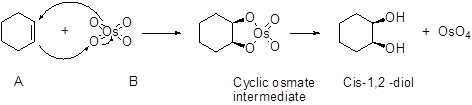
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What products are obtained from the reaction of the following compounds with H2CrO4 + ∆?
What products are obtained from the reaction of the following compound with H2CrO4 + Δ?
Draw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2
Chapter 10 Solutions
Modified Mastering Chemistry with Pearson eText -- Standalone Access Card -- for Organic Chemistry (8th Edition)
Ch. 10.1 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 10.1 - Explain the difference in reactivity between...Ch. 10.1 - Prob. 5PCh. 10.1 - Prob. 6PCh. 10.1 - Prob. 8PCh. 10.2 - Prob. 9PCh. 10.3 - Prob. 11PCh. 10.3 - Show how 1-propanol can be converted into the...Ch. 10.4 - Which of the following alcohols dehydrates the...Ch. 10.4 - Prob. 14P
Ch. 10.4 - Prob. 15PCh. 10.4 - Propose a mechanism for each of the following...Ch. 10.4 - Draw the product of each of the following...Ch. 10.4 - Explain why the following alcohols, when heated...Ch. 10.4 - What stereoisomers are formed in the following...Ch. 10.4 - Prob. 20PCh. 10.4 - What alcohol would you treat with phosphorus...Ch. 10.5 - Prob. 22PCh. 10.6 - What are the major products obtained when each of...Ch. 10.6 - Prob. 26PCh. 10.7 - Prob. 27PCh. 10.7 - Would you expect the reactivity of a five-membered...Ch. 10.7 - Prob. 29PCh. 10.7 - What products are obtained from the reaction of...Ch. 10.7 - Prob. 31PCh. 10.7 - Prob. 32PCh. 10.7 - Prob. 33PCh. 10.8 - Draw the mechanism for formation of the two...Ch. 10.8 - Prob. 35PCh. 10.8 - Prob. 36PCh. 10.8 - How do the major products obtained from...Ch. 10.8 - Explain why the two arene oxides in Problem 38...Ch. 10.8 - Which compound is more likely to be carcinogenic?Ch. 10.8 - Three arene oxides can be obtained from...Ch. 10.9 - Explain why the half-life (the time it takes for...Ch. 10.10 - Prob. 43PCh. 10.10 - Prob. 44PCh. 10.10 - Prob. 45PCh. 10.10 - Prob. 46PCh. 10.10 - Prob. 47PCh. 10.10 - Describe a synthesis for each of the following...Ch. 10.11 - Using an alkyl halide and a thiol as starting...Ch. 10.11 - The following three nitrogen mustards were studied...Ch. 10.11 - Why is melphalan a good cancer drug?Ch. 10.11 - Prob. 53PCh. 10.12 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 55PCh. 10 - Which compound is more likely to be carcinogenic?Ch. 10 - Prob. 57PCh. 10 - Prob. 58PCh. 10 - When heated with H2SO4, both...Ch. 10 - What is the major product obtained from the...Ch. 10 - Write the appropriate reagent over each arrow.Ch. 10 - What alkenes would you expect to be obtained from...Ch. 10 - Prob. 63PCh. 10 - Prob. 64PCh. 10 - When deuterated phenanthrene oxide undergoes a...Ch. 10 - An unknown alcohol with a molecular formula of...Ch. 10 - Explain why the acid-catalyzed dehydration of an...Ch. 10 - Prob. 68PCh. 10 - Prob. 69PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - What product would be formed if the four-membered...Ch. 10 - Which of the following ethers would be obtained in...Ch. 10 - Using the given starting material any necessary...Ch. 10 - Prob. 74PCh. 10 - When 3-methyl-2-butanol is heated with...Ch. 10 - Draw structures for compounds AF.Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - How could you synthesize isopropyl propyl ether,...Ch. 10 - When ethyl ether is heated with excess HI for...Ch. 10 - When the following seven-membered ring alcohol is...Ch. 10 - Ethylene oxide reacts readily with HO because of...Ch. 10 - Describe how each of the following compounds could...Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - Triethylene glycol is one of the products obtained...Ch. 10 - Prob. 85PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 87PCh. 10 - An ion with a positively charged nitrogen atom in...Ch. 10 - The following reaction takes place several times...Ch. 10 - Prob. 90PCh. 10 - Propose a mechanism for each of the following...Ch. 10 - A vicinal diol has OH groups on adjacent carbons....Ch. 10 - Prob. 93PCh. 10 - Prob. 94PCh. 10 - Two stereoisomers are obtained from the reaction...Ch. 10 - Propose a mechanism for each or the following...Ch. 10 - Triethylenemelamine (TEM) is an antitumor agent....
Knowledge Booster
Similar questions
- Draw the products formed when the following alkynes are treated with each set of reagents: [1] H2O, H2SO4, HgSO4; or [2] R2BH followed by H2O2, −OH.arrow_forwardWhat products are obtained from metathesis of each of the following alkenes?arrow_forwardWhat is the major product obtained from the reaction of 2-ethyloxirane with each of the following reagents? a. 0.1 M HCl b. CH3OH>HCl c. 0.1 M NaOH d. CH3OH/CH3O-arrow_forward
- Predict the products of reaction of pent-1-yne with the following reagents. H2SO4>HgSO4, H2Oarrow_forwardCompeting SN and E reactions. Predict whether the following reactions will proceed via substitution (SN1 or SN2), or elimination (E₁ or E₂), or whether the two will compete. Identify the major product.arrow_forwardWhat will be the major product of the reaction of 2-methyl-2-butene with each of the following reagents? a) Br2/H2O b)BH3/THF followed by H2O/Hydeoxyl ionarrow_forward
- What is the major substitution product for the following reaction? Show the mechanism for the reaction.arrow_forwardWhat is the major product obtained from the acid-catalyzed hydration of each of the following alkenes?arrow_forwardDraw the major product(s) obtained from the acid-catalyzed hydration of the following alkene: CH3CH2CH2CH=CH2arrow_forward
- Predict the products of the reaction with benzaldehyde with the following reagents? NaBH4, then H3O+ Dess_Martin reagent Methylmagnesium bromide, then H3O+ (C6H5)3P=CH2arrow_forwardwhat products are formed from reaction of HBr with racemic 4-Methyl-1-hexenearrow_forwardWhat is the major product of the reaction of 1 mol of propyne with each of the following reagents? a. HBr (1 mol) e. aqueous H2SO4, HgSO4 h. H2/Lindlar catalyst b. HBr (2 mol) f. R2BH in THF followed by i. sodium amide c. Br2 (1 mol)/CH2Cl2 H2O2/HO-/H2O j. the product of part i followed by d. Br2 (2 mol)/CH2Cl2 g. excess H2, Pd/C 1-chloropropanearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning