
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
9th Edition
ISBN: 9781292151229
Author: Wade, LeRoy G.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10.9D, Problem 10.18P
A formate ester, such as ethyl formate, reacts with an excess of a Grignard reagent to give (after protonation) secondary alcohols with two identical alkyl groups.
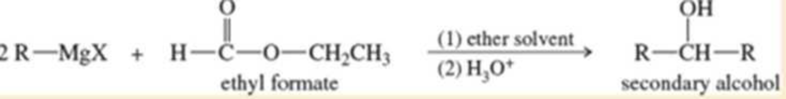
- a. Propose a mechanism to show how the reaction of ethyl formate with an excess of allylmagnesium bromide gives, after protonation, hepta-1,6-dien-4-ol.

- b. Show how you would use reactions of Grignard reagents with ethyl formate to synthesize the following secondary alcohols.
- i. pentan-3-ol
- ii. diphenylmethanol
- iii. trans,trans-nona-2,7-dien-5-ol
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What products are obtained from the following reactions?
a. ethyl benzoate + excess isopropanol + HCl b. phenyl acetate + excess ethanol + HCl
Which of the provided schemes can be used to
synthesize p-chlorophenol
p-chlorophenol from benzen
e?
A. None of the below schemes are
correct
B.
O C.
D.
SO3
H₂SO4
Cl₂
FeCl
903
H₂SO4
Product
Product
Product
1. NaOH
2. H₂O*
SO 3
H₂SO4
Cl₂
FeCl
Product
Product
Product
Cl₂
FeCl
H₂/Pd
1. NaOH
2. H₂O*
p-Chlorophenol
p-Chlorophenol
p-Chlorophenol
Choose the reagent(s) that would be most likely to
complete this reaction.
HILL
Y....
A
B
C
D
E
BH3-THF
H2O2, NaOH
tBuOK
CHCI 3
CH2I2
Zn/Cu
mCPBA
1. Hg(OAc)2, H₂O
2. NaBH4, NaOH
Chapter 10 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
Ch. 10.3A - Prob. 10.1PCh. 10.3B - Give both the IUPAC name and the common name for...Ch. 10.3B - Prob. 10.3PCh. 10.3C - Give a systematic (IUPAC) name for each diol. a....Ch. 10.4B - Predict which member of each pair will be more...Ch. 10.4B - Dimethylamine (CH3)2NH, has a molecular weight of...Ch. 10.6A - Prob. 10.7PCh. 10.6A - Prob. 10.8PCh. 10.6C - A nitro group (NO2) effectively stabilizes a...Ch. 10.6C - Prob. 10.10P
Ch. 10.8B - Prob. 10.11PCh. 10.8B - Prob. 10.12PCh. 10.9A - Prob. 10.13PCh. 10.9B - Prob. 10.14PCh. 10.9C - Show how you would synthesize each tertiary...Ch. 10.9D - Prob. 10.16PCh. 10.9D - Show how you would add Grignard reagents to acid...Ch. 10.9D - A formate ester, such as ethyl formate, reacts...Ch. 10.9E - Prob. 10.19PCh. 10.9E - In Section9-7B, we saw how acetylide ions add to...Ch. 10.9F - Prob. 10.21PCh. 10.10A - Prob. 10.22PCh. 10.10B - Prob. 10.23PCh. 10.11B - Predict the products you would expect from the...Ch. 10.11B - Prob. 10.25PCh. 10.11B - Prob. 10.26PCh. 10.12 - Prob. 10.27PCh. 10.12 - Prob. 10.28PCh. 10.12 - Authentic skunk spray has become valuable for use...Ch. 10 - Give a systematic (IUPAC) name for each alcohol....Ch. 10 - Give systematic (IUPAC) names for the following...Ch. 10 - Draw the structures of the following compounds...Ch. 10 - Predict which member of each pair has the higher...Ch. 10 - Predict which member of each pair is more acidic,...Ch. 10 - Predict which member of each group is most soluble...Ch. 10 - Draw the organic products you would expect to...Ch. 10 - Prob. 10.37SPCh. 10 - Show how you would synthesize the following...Ch. 10 - Show how you would use Grignard syntheses to...Ch. 10 - Show how you would accomplish the following...Ch. 10 - Show how you would synthesize the following: a....Ch. 10 - Complete the following acid-base reactions. In...Ch. 10 - Prob. 10.43SPCh. 10 - Prob. 10.44SPCh. 10 - Geminal diols, or 1,1-diols, are usually unstable,...Ch. 10 - Vinyl alcohols are generally unstable, quickly...Ch. 10 - Compound A (C7H11Br) is treated with magnesium in...Ch. 10 - Prob. 10.48SPCh. 10 - Prob. 10.49SPCh. 10 - Prob. 10.50SPCh. 10 - Prob. 10.51SPCh. 10 - Prob. 10.52SPCh. 10 - Prob. 10.53SPCh. 10 - Prob. 10.54SPCh. 10 - Prob. 10.55SPCh. 10 - Prob. 10.56SPCh. 10 - Show how this 1 alcohol can be made from the...Ch. 10 - Prob. 10.58SPCh. 10 - Prob. 10.59SPCh. 10 - Prob. 10.60SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Provide the product most likely to form from reaction of each reagent with 1-methylcyclohexene. You may ingore stereochemistry for this question. Br2 H-Br H-Br, PhC(O)OOC(O)Ph N-bromosuccinimide, PhC(O)OOC(O)Ph A. B. C. D.arrow_forwardWhat is the major product of the reaction of 1 mol of propyne with each of the following reagents? a. HBr (1 mol) e. aqueous H2SO4, HgSO4 h. H2/Lindlar catalyst b. HBr (2 mol) f. R2BH in THF followed by i. sodium amide c. Br2 (1 mol)/CH2Cl2 H2O2/HO-/H2O j. the product of part i followed by d. Br2 (2 mol)/CH2Cl2 g. excess H2, Pd/C 1-chloropropanearrow_forward3. Predict the major product and provide a mechanism for the following: 1-(cyclopentyl-1-ol)-cyclopentanol and sulfuric acid a. b. 2-methyl-1,2-butanediol and sulfuric acid c. 3-cyclopentyl-1,2-propanediol and sulfuric acidarrow_forward
- What is the best set of reagents to achieve deoxygenation of 2-pentanone to pentane? A. NaClO2/NaH2PO4 B. LiAlH4, Et2O C. DIBAL-H, THF D. NH2NH2/t-BuOK, DMSOarrow_forwardReagents CH3 a. HNO3, H2SO4 f. CH3OH LOCH3 b. SnCl2, H3O+ then NaOH(aq) g. CuCN C. HNO2, HCI h. Nal d. HBF4 i. LIAIH4 then H₂O e. KMnO4 j. CH3COCI Choose reagents from the table to synthesize this compound from toluene. Enter the letters in the order that you wish to use the reagents, without spaces or punctuation, i.e. 'adeg'. Submit Answer Try Another Version 2 item attempts remaining Visitedarrow_forwardC D and F pleasearrow_forward
- 17. Provide the reagents necessary to carry out the 18. Predict the product(s) for the following conversion. following reaction. A. B. C. D. E. NaBH4/CH3OH Na/NH3 1. 2. LIAIH4 H₂O H3O+/heat A and C OH C(CH₂) 141 CH₂OC(CH₂) 14CH3 HOOL CHOC(CH2) 16CH3 CH₂OC(CH2) 14CH3 (CH₂)14C NaOH excess Aarrow_forward2. Provide either the reagents or the major organic product of each reaction, using reactions and reagents we have studied in this course One step A. B. G D. Br =H 1.03 2. Zn, acetic acid reagents?? Cl₂ (=arrow_forward4. i. Define Hemiacetal, Acetal and Ketal. ii. Give the structure of the product from the reaction of propanal with 1M ethanol in dry acid. iii. What happens when Further 1M of ethanol is added to above?arrow_forward
- Which is the best method for the following transformation? OH A. 1. BH3, THF 2. H₂O₂, OH- B. H¹, H₂O C. concentrated H₂SO4 D. 1. Hg(OAc)2, H₂O 2. NaBH4arrow_forwardSynthesize the following compound from cyclohexanol using any other organic or inorganic compounds. HO. Part 1 out of 2 Preparation of the Grignard reagent: Book rint rences OH draw structure ... draw structure... Draw the intermediate product formed above and select the correct reagent A. O MgBr, O CH;Br O Brz PB13 Draw the intermediate product formed above and select the correct reagent B. O MgBr O Mgo O O Mgl, O Mg Hint Prev 3 of .8 Next > re to searcharrow_forwardIndicate how the following compounds can be synthesized from cyclohexanone and any other necessary reagents:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY