
Concept explainers
a)
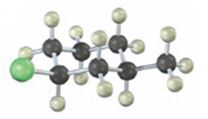
Interpretation:
The IUPAC name of the
Concept introduction:
The longest continuous carbon chain in the molecule is chosen. The chain is numbered from the end which gives lowest number to the substituent either halo or alkyl group present. If different halogens are present they are numbered and listed in the alphabetical order while writing the name. If same two alternatives exist for different substituents, then the chain is numbered from the end that gives lowest number to the substituent that has alphabetical preference.
To give:
The IUPAC name of the alkyl halide shown.
Answer to Problem 14VC
The alkyl halide given is
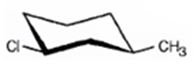
Its IUPAC name is cis- 1- chloro-3-methylcyclohexane.
Explanation of Solution
The compound has a cyclohexane ring with a Cl atom on C1 and a methyl on C3 both at equatorial positions. Hence its name is cis- 1- chloro-3-methylcyclohexane.
The IUPAC name of the alkyl halide shown is cis- 1- chloro-3-methylcyclohexane.
b)
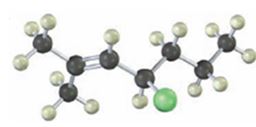
Interpretation:
The IUPAC name of the alkyl halide is to be given.
Concept introduction:
The longest continuous carbon chain in the molecule is chosen. The chain is numbered from the end which gives lowest number to the substituent either halo or alkyl group present. If different halogens are present they are numbered and listed in the alphabetical order while writing the name. If same two alternatives exist for different substituents, then the chain is numbered from the end that gives lowest number to the substituent that has alphabetical preference.
To give:
The IUPAC name of the alkyl halide shown.
Answer to Problem 14VC
The alkyl halide given is
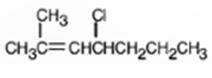
Its IUPAC name is 4-chloro-2-methyl-2-heptene.
Explanation of Solution
The compound has a seven carbon straight chain with a double bond between C2 & C3, a Cl atom on C4 and a methyl on C3. Hence its name is 4-chloro-2-methyl-2-heptene.
The IUPAC name of the alkyl halide shown is 4-chloro-2-methyl-2-heptene.
Want to see more full solutions like this?
Chapter 10 Solutions
Bundle: Organic Chemistry, Loose-Leaf Version, 9th + OWLv2, 1 term (6 months) Printed Access Card
- Draw the products, including their configurations, obtained from the reaction of 1-ethylcyclohexene with the following reagents:a. HBr b. H2, Pd/C c. R2BH/THF, followed by HO– , H2O2, H2O d. Br2/CH2Cl2arrow_forwardDraw the structure of the four allylic halides formed when 3-methylcyclohexene undergoes allylic halogenation with NBS + hv.arrow_forward1. Predict the products, if any, for the reaction of benzene with the following reagents: a. Br2/CCl4, room temperature b. C2H5Cl, AlCl3arrow_forward
- 1. Ethylene glycol, a major component of antifreeze, becomes especially poisonous when it is oxidized by the liver. Draw two oxidation products that could be derived from ethylene glycol. 2.When 2-methyl-1-butanol is dehydrated in an acid medi- um to an alkene, it yelds mainly 2-methyl-2-butene rather than 2-methyl-1-butene. This indicates that the dehydra- tion to an alkene is at least a two-step reaction. Suggest a mechanism to explain the reaction.arrow_forwardWhat is the product of the reaction of 2-methyl-2-butene with Br2/H2O? 3-bromo-2-methyl-2-butanol 2-bromo-3-methyl-2-butanol 4-bromo-2-methyl-2-butanol 2,3-dibromo-2-methylbutanearrow_forwardGive IUPAC names for the following substances (red = O, blue = N):arrow_forward
- Another important pattern in organic synthesis is the construction of CC bonds. Using your reaction roadmap as a guide, show how to convert propane into hex-1-en-4-yne. You must use propane as the source of all of the carbon atoms in the hex-1-en-4-yne product. Show all reagents needed and all molecules synthesized along the way.arrow_forwardAlcohols can be made by reacting alkyl halides with sodium hydroxide as follows:RX +. NaOH ROH + NaX Give the names and formulas of the alcohols produced from the following alkyl bromides by this method:(a) 2-bromobutane(b) 2-bromo-3-ethylpentane(c) bromocyclopentanearrow_forwardThe pictured reaction shows an alkyl bromide being converted into an alkene. Choose all reagents that would produce the pictured alkene as the major product. A) NaOH/H2O B) H2O C) tBuOK/tBuOH D) EtONa/EtOHarrow_forward
- 1. Write reactions of butane with the following reagents: a. Br2/t °C; b. KMnO4/ H2O/20 °C;arrow_forwardDescribe how 1-ethylcyclohexanol can be prepared from cyclohexane. You can use any inorganic reagents, any solvents, and any organic reagents as long as they contain no more than two carbons.arrow_forwardWrite reactions of 2-methyl propane with the following reagents: b. SO2 + Cl2°C; c. HNO3 dil., t °C, p.arrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


