
Concept explainers
Draw structures for the three
Interpretation: The three alkynes having molecular formula C5H8 is to be drawn and each is to be classified as an internal or terminal alkyne.
Concept Introduction: Alkynes are an unsaturated hydrocarbon having at least one carbon-carbon triple bond. The general formula of alkynes are CnH2n-2. The simplest hydrocarbon of alkyne family is acetylene having two carbon atom. Alkynes can be divided into the following two categories based on the position of triple bond; 1) internal alkyne 2) terminal alkyne. In internal alkynes there are no hydrogen atoms bonded to triply bonded carbon atoms. In terminal alkynes there is at least one hydrogen atom bonded to a triply bonded carbon atom.
Answer to Problem 11.1P
the internal and terminal alkynes of molecular formula C5H8 are as follows;
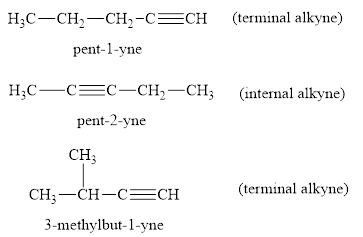
Explanation of Solution
In pent-1-yne and 3-methyl but-1-yne the triple bond is at the terminal side of carbon having one -C-H group. Thus, they are called as terminal alkynes. In case of pent-2-yne the triple bond is in the middle of carbon having no free -C-H group. Thus, it is called as internal alkyne.
Thus, the alkynes of formula C5H is drawn and named as an internal alkyne or terminal alkyne.
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry - With Access (Custom)
- draw structural formulas for all isomeric alkanes with molecular formula C7H16. Predict which isomer has the lowest boiling point and which has the highest boiling point.arrow_forwardClassify alkyl halides as primary, secondary, tertiary, aliphatic, or benzenearrow_forwardA hydrocarbon C5H12 gives only one mono-chlorination product. Identify the hydrocarbon.arrow_forward
- There are 18 isomeric alkanes of molecular formula C8H18. Draw and name any eight of them.arrow_forwardName the cycloalkanes with molecular formula C6H12 that have a 3-membered ring and two substituents.arrow_forwardDescribe each highlighted bond in terms of the overlap of atomic orbitals. and Draw the structures of ALL of the aldehydes with the molecular formula C5H10O that contain a 5-carbon chain.arrow_forward
- Draw the structure of the aldehydes with molecular formula C5H10O that contain an unbranched carbon chain.arrow_forwardWrite structural formulas for all aldehydes with the molecular formula C6H12O and give each its IUPAC name. Which of these aldehydes are chiralarrow_forwardWrite a conformational structure for 1,2,3-trimethylcyclohexane in which all the methyl groups are axial and then show its more stable conformation.arrow_forward
- Name the 7 aldehydes and ketones with formulaC5H10Oarrow_forwardThe sex attractant of the female tiger moth is an alkane of molecular formula C 18H 38. Is this molecule an acyclic alkane or a cycloalkane?arrow_forwardHow many ethers have molecular formula C5H12O? Draw their structures and give each a systematic name. What are their common names?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

