
Concept explainers
(a)
Interpretation:
The condensed formula of
Concept Introduction:
Condensed structural formula: Condensed structural formula shows the arrangement of atoms in grouped form.
(a)
Answer to Problem 11A.12E
The condensed formula of
Explanation of Solution
The given compound is,
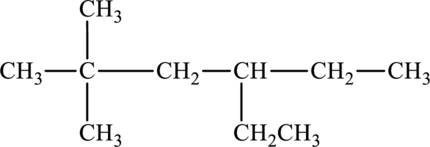
The parent chain of the compound is hexane. One ethyl group is present in the carbon fourth position and two methyl groups are present at carbon second position. Hence, the systematic name of the compound is
(b)
Interpretation:
The condensed formula of
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 11A.12E
The condensed formula of
Explanation of Solution
The given compound is,
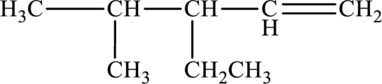
The parent chain of the compound is pentane. One ethyl group is present in the carbon third position and one methyl group is present in carbon fourth position. One double bond is present at the carbon first position.
Hence, the systematic name of the compound is
(c)
Interpretation:
The condensed formula of
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 11A.12E
The condensed formula of
Explanation of Solution
The given compound is,
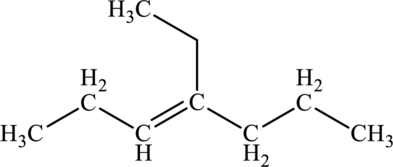
The parent chain of the compound is heptane. One methyl group is present in carbon fourth position and a double bond is seen at the carbon third position. Hence, the systematic name of the compound is
(d)
Interpretation:
The condensed formula of
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 11A.12E
The condensed formula of
Explanation of Solution
The given compound is,
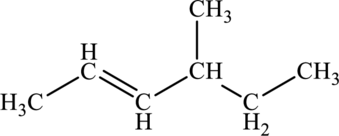
The parent chain of the compound is hexane. One methyl group is present in the carbon fourth position and a double bond is present in carbon second position.
Hence, the systematic name of the compound is
Want to see more full solutions like this?
Chapter 11 Solutions
EBK CHEMICAL PRINCIPLES
- Give the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardWrite the chemical equation for the reaction of propanoic acid with 1-butanol (an alcohol). The formula of 1-butanol is CH-CH-CH2-CH2-OH. Which is formula of the this rule of reaction?arrow_forwarddraw the structure of C8H10NO7P2- and give the general name of the functional grouparrow_forward
- Draw and name the five cycloalkane structures of formula C5H10. Can any of these structures give rise to geometric (cis-trans) isomerism? If so, show the cis and trans stereoisomersarrow_forwardWhat is an isomer having the formula C6H12 ?Without using 2-hexenearrow_forward(a) What is meant by the term isomer ? (b) Among the four alkanes, ethane, propane, butane, and pentane, which is capable of existing in isomeric forms?arrow_forward
- Write the suffix -ene for alkenes and -yne for alkynesarrow_forwardwrite the structure formulas of alkanes with molecular formula C6H14, which with chlorine give: a) three monochlorinated isomers? b) five monochlorinated isomers c) only two monochlorinated isomersarrow_forwardDraw the structures of the 2 isomers of C8H18 that contain 2 methyl branches on the same carbon of the main chain.arrow_forward
- Write any two structural isomers of C5H12?arrow_forwardWhat structural features help us identify a compound as(a) an alkane, (b) a cycloalkane, (c) an alkene, (d) an alkyne,(e) a saturated hydrocarbon, (f) an aromatic hydrocarbon?arrow_forwardGive the molecular formula of a hydrocarbon containingfive carbon atoms that is (a) an alkane, (b) a cycloalkane,(c) an alkene, (d) an alkyne.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




