
(a)
Interpretation:
It has to be shown that the reaction obeys the rate law as given below.
Concept Introduction:
In a first-order reaction, the graph of concentration vs. rate is linear.
(a)
Explanation of Solution
Average concentrations can be calculated as given below.
From
From
From
From
From
From
The average rate and concentration is given in the table below.
| Average concentration ( | Average rate ( |
A graph of concentration vs. rate is given below.
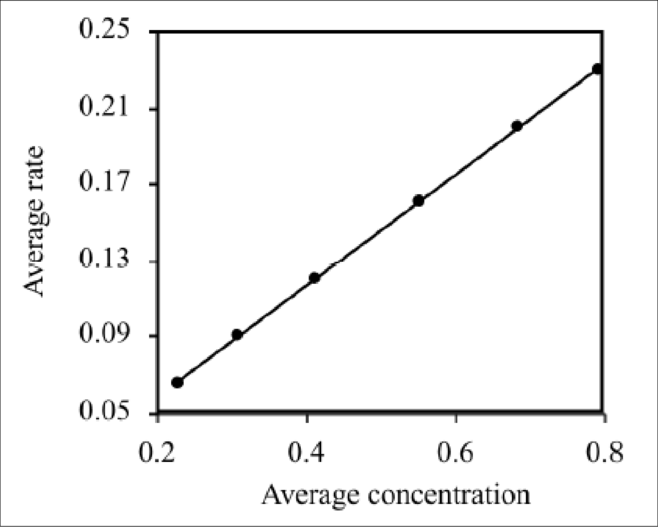
Figure 1
The above graph is linear which satisfies the rate law.
(b)
Interpretation:
The rate constant
(b)
Answer to Problem 14QRT
The average value of
Explanation of Solution
The rate constant
From
Average rate is
Average concentration is
Then, the rate constant can be calculated as given below.
From
Average rate is
Average concentration is
Then, the rate constant can be calculated as given below.
From
Average rate is
Average concentration is
Then, the rate constant can be calculated as given below.
From
Average rate is
Average concentration is
Then, the rate constant can be calculated as given below.
From
Average rate is
Average concentration is
Then, the rate constant can be calculated as given below.
From
Average rate is
Average concentration is
Then, the rate constant can be calculated as given below.
The average value of
(c)
Interpretation:
The
(c)
Answer to Problem 14QRT
The reaction rate exactly
Explanation of Solution
The initial concentration of
The rate law describes that the reaction is a first-order reaction. The integrated first-order rate law is given below.
Now, the rate of the reaction at
Therefore, the reaction rate exactly
Want to see more full solutions like this?
Chapter 11 Solutions
EBK CHEMISTRY: THE MOLECULAR SCIENCE
- What will the units of the rate constants in Question 12 be if the rate is expressed in mol/L min?arrow_forwardUnder certain conditions the decomposition of ammonia on a metal surface gives the following data: [NH3] (M) 1.0103 2.0103 3.0103 Rate (moI/L/h1) 1.5106 1.5106 1.5106 Determine the rate equation, the rate constant, and the overall order for this reaction.arrow_forwardThe decomposition of iodoethane in the gas phase proceeds according to the following equation: C2H5I(g)C2H4(g)+HI(g) At 660. K, k = 7.2 104 sl; at 720. K, k = 1.7 102 sl. What is the value of the rate constant for this first-order decomposition at 325C? If the initial pressure of iodoethane is 894 torr at 245C, what is the pressure of iodoethane after three half-lives?arrow_forward
- The half-life of tritium, 3H, is 12.26 years. Tritium is the radioactive isotope of hydrogen. (a) What is the rate constant for the radioactive decay of tritium, in y1 and s1? (b) What percentage of the original tritium is left after 61.3 years?arrow_forwardThe compound Xe(CF3)2 decomposes in a first-order reaction to elemental Xe with a half-life of 30. minutes. If you place 7.50 mg of Xe(CF3)2 in a flask, how long must you wait until only 0.25 mg of Xe(CF3)2 remains?arrow_forwardWhen formic acid is heated, it decomposes to hydrogen and carbon dioxide in a first-order decay. HCOOH(g) CO2(g) + H2(g) At 550 C, the half-life of formic acid is 24.5 minutes. (a) What is the rate constant, and what are its units? (b) How many seconds are needed for formic acid, initially 0.15 M, to decrease to 0.015 M?arrow_forward
- The following rate constants were obtained in an experiment in which the decomposition of gaseous N2O; was studied as a function of temperature. The products were NO, and NO,. Temperature (K) 3.5 x 10_i 298 2.2 x 10"4 308 6.8 X IO-4 318 3.1 x 10 1 328 Determine Etfor this reaction in kj/mol.arrow_forwardSuppose that the half-life of steroids taken by an athlete is 42 days. Assuming that the steroids biodegrade by a first-order process 5, how long would it take for 164 of the initial dose to remain in the athlete’s body?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





