
CHEMISTRY THE CENTRAL SCIENCE 14TH EDI
14th Edition
ISBN: 9780134863016
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11, Problem 4E
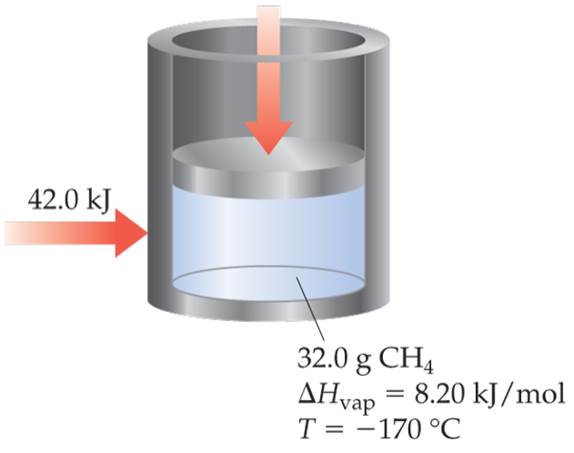
If 42.0 kj of heat is added to a 32.0-g sample of liquid methane under 1 atm of pressure at a temperature of - 170ºC, what are the final state and themperature of the methane once the system equilibrates? Assume no heat is lost to the surroundings. The normal boiling point of methane is -161.5ºC.The specific heats of liquid and gaseous methane are 3.48 and 2.22 J/g-K, respectively. [Section 11.4□]
P= 1.00 atm
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
CHEMISTRY THE CENTRAL SCIENCE 14TH EDI
Ch. 11.2 - Which of the following substances is most likely...Ch. 11.2 - Prob. 11.1.2PECh. 11.3 - Prob. 11.2.1PECh. 11.3 - Prob. 11.2.2PECh. 11.4 - What information about water is needed to...Ch. 11.4 - Prob. 11.3.2PECh. 11.5 -
In the mountains, water in an open container will...Ch. 11.5 - Prob. 11.4.2PECh. 11.6 - Prob. 11.5.1PECh. 11.6 - Prob. 11.5.2PE
Ch. 11.7 - Liquid crystalline phases are produced by which of...Ch. 11.7 - Prob. 11.6.2PECh. 11 - Prob. 1DECh. 11 - Prob. 1ECh. 11 - Prob. 2ECh. 11 - Prob. 3ECh. 11 - If 42.0 kj of heat is added to a 32.0-g sample of...Ch. 11 - Prob. 5ECh. 11 - The molecules have the same molecular formula...Ch. 11 - Prob. 7ECh. 11 - Prob. 8ECh. 11 - Prob. 9ECh. 11 - Prob. 10ECh. 11 - Prob. 11ECh. 11 - Prob. 12ECh. 11 - Prob. 13ECh. 11 - Prob. 14ECh. 11 - Prob. 15ECh. 11 - Prob. 16ECh. 11 - Describe the intermolecular forces that must be...Ch. 11 - Prob. 18ECh. 11 - List the following molecules in order of...Ch. 11 - True or false: a. For molecules with similar...Ch. 11 - Prob. 21ECh. 11 - Prob. 22ECh. 11 - Prob. 23ECh. 11 - Prob. 24ECh. 11 - Prob. 25ECh. 11 - Prob. 26ECh. 11 - Prob. 27ECh. 11 - Prob. 28ECh. 11 - Prob. 29ECh. 11 - Prob. 30ECh. 11 - A number of salts containing the tetrahedral...Ch. 11 - Prob. 32ECh. 11 - a. What is the relationship between surface...Ch. 11 - Prob. 34ECh. 11 - Prob. 35ECh. 11 - Prob. 36ECh. 11 - The boiling points, surface tension, and...Ch. 11 - Prob. 38ECh. 11 - Prob. 39ECh. 11 - Prob. 40ECh. 11 - Prob. 41ECh. 11 - Prob. 42ECh. 11 - 11.43 For many years drinking water has been...Ch. 11 - Prob. 44ECh. 11 - Prob. 45ECh. 11 - The fluorocarbon compound C2 Cl 3£'y has a normal...Ch. 11 - 11.47 Indicate whether each statement is true or...Ch. 11 - Prob. 48ECh. 11 - 11,49 Which of the following affects the vapor...Ch. 11 - Prob. 50ECh. 11 - Prob. 51ECh. 11 - Prob. 52ECh. 11 - Prob. 53ECh. 11 - Prob. 54ECh. 11 - Prob. 55ECh. 11 - Prob. 56ECh. 11 - Prob. 57ECh. 11 - Prob. 58ECh. 11 - Prob. 59ECh. 11 - Prob. 60ECh. 11 - Prob. 61ECh. 11 - Prob. 62ECh. 11 - Prob. 63ECh. 11 - Prob. 64ECh. 11 - In terms of the arrangement and freedom of motion...Ch. 11 - Prob. 66ECh. 11 - Prob. 67ECh. 11 - Prob. 68ECh. 11 - Prob. 69ECh. 11 - Prob. 70ECh. 11 - Prob. 71ECh. 11 - Prob. 72ECh. 11 - Prob. 73AECh. 11 - Prob. 74AECh. 11 - Prob. 75AECh. 11 - Prob. 76AECh. 11 - Prob. 77AECh. 11 - The table below shows the normal boiling points of...Ch. 11 - Prob. 79AECh. 11 - Prob. 80AECh. 11 - Prob. 81AECh. 11 - Prob. 82AECh. 11 - Prob. 83AECh. 11 - Prob. 84AECh. 11 - Prob. 85AECh. 11 - Prob. 86AECh. 11 - Prob. 87AECh. 11 - Prob. 88AECh. 11 - Prob. 89AECh. 11 - Prob. 90IECh. 11 - Prob. 91IECh. 11 - Prob. 92IECh. 11 - 11.93 The vapor pressure of ethanol (C2H5OH) at 19...Ch. 11 - Prob. 94IECh. 11 - Prob. 95IECh. 11 - Prob. 96IECh. 11 - Prob. 97IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- he enthalpy (H)of vaporization of water is about seven times larger than water’s enthalpy fusion(41kJ/molvs.6kJ/mol). What does this tell us about the relative similarities among the solid, liquid, and gaseous states of water?arrow_forwardWhat is U when 1.00 mol of liquid water vaporizes at 100C? The heat of vaporization, Hvap, of water at 100C is 40.66 kJ/mol.arrow_forwardLiquid butane, C4H10, is stored in cylinders to be used as a fuel. Suppose 35.5 g of butane gas is removed from a cylinder. How much heat must be provided to vaporize this much gas? The heat of vaporization of butane is 21.3 kJ/mol.arrow_forward
- 5-86 Using the phase diagram of water (Figure 5-20), describe the process by which you can sublime 1 g of ice at-10°C and at 1 atm pressure to water vapor at the same temperature.arrow_forwardA 0.250-g chunk of sodium metal is cautiously dropped into a mixture of 50.0 g water and 50.0 g ice, both at 0C. The reaction is 2Na(s)+2H2O(l)2NaOH(aq)+H2(g)H=368kJ Assuming no heat loss to the surroundings, will the ice melt? Assuming the final mixture has a specific heat capacity of 4.18 J/gc, calculate the final temperature. The enthalpy of fusion for ice is 6.02 kJ/mol.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Viscosity, Cohesive and Adhesive Forces, Surface Tension, and Capillary Action; Author: Professor Dave Explains;https://www.youtube.com/watch?v=P_jQ1B9UwpU;License: Standard YouTube License, CC-BY