
Conceptual Integrated Science
3rd Edition
ISBN: 9780135197394
Author: Hewitt, Paul G., LYONS, Suzanne, (science Teacher), Suchocki, John, Yeh, Jennifer (jennifer Jean)
Publisher: PEARSON EDUCATION (COLLEGE)
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11, Problem 74TE
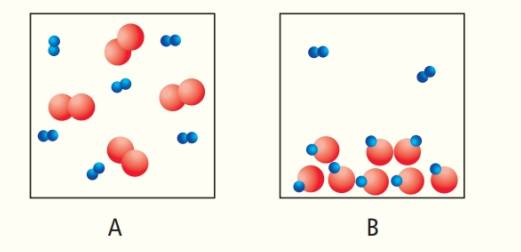
Each sphere in the diagrams shown here represents an atom. Joined spheres represent molecules. Which box contains a liquid phase? Why can you not assume that box B represents a lower temperature?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What does it mean for water to be a polar molecule?
Why is the fact that ice is less dense than liquid water so unusual?
What are the benefits of the high specific heat of water?
Complete the table below to answer the question How do particles of solid, liquid and gas differ?
Directions: Analyze the illustration and answer the given questions below
In what phase is the substance at 250 K and 50 atm pressure?
What phases will exist at 1 atm and 195.5 K?
What is the significance of pressure 5.2 atm?
Describe the changes that take place when a sample of solid CO2 is heated at a pressure of 1 atm, starting at a temperature of 195.5K
Chapter 11 Solutions
Conceptual Integrated Science
Ch. 11 - Prob. 1RCCCh. 11 - Prob. 2RCCCh. 11 - Prob. 3RCCCh. 11 - Prob. 4RCCCh. 11 - Prob. 5RCCCh. 11 - How are the particles in a solid arranged...Ch. 11 - Which occupies the greatest volume: 1 gram of ice,...Ch. 11 - What is it called when evaporation takes place...Ch. 11 - How is sublimation different from evaporation?Ch. 11 - Prob. 10RCC
Ch. 11 - How much heat is needed to melt 1 gram of ice?...Ch. 11 - What happens to the chemical identity of a...Ch. 11 - What is a physical property? A chemical property?Ch. 11 - What is a chemical bond?Ch. 11 - What changes during a chemical reaction?Ch. 11 - Why is the freezing of water considered to be a...Ch. 11 - Prob. 17RCCCh. 11 - Why is the rusting of iron considered to be a...Ch. 11 - Prob. 19RCCCh. 11 - What is the difference between an element and a...Ch. 11 - How many atoms are in one molecule of H3PO4?Ch. 11 - How many atoms of each element are in one molecule...Ch. 11 - What does the chemical formula of a substance tell...Ch. 11 - Prob. 24RCCCh. 11 - Prob. 25RCCCh. 11 - Prob. 26RCCCh. 11 - What is the chemical formula for the compound...Ch. 11 - Why are common names often used for chemical...Ch. 11 - How soon will nanotechnology give rise to...Ch. 11 - Prob. 30TISCh. 11 - Who is the ultimate expert at nanotechnology?Ch. 11 - Prob. 38TCCh. 11 - Rank these substances in order of increasing...Ch. 11 - Rank the following physical and chemical changes...Ch. 11 - Rank these compounds in order of increasing number...Ch. 11 - How has chemistry influenced our modern...Ch. 11 - While visiting a foreign country, a foreign...Ch. 11 - If someone is able to explain an idea to you using...Ch. 11 - What is the best way to really prove to yourself...Ch. 11 - Prob. 46TECh. 11 - Prob. 47TECh. 11 - What is found between two adjacent molecules of a...Ch. 11 - You combine 50mL of water with 50mL of purified...Ch. 11 - Prob. 50TECh. 11 - Which has stronger attractions among its...Ch. 11 - Prob. 52TECh. 11 - Is it possible for air to be in liquid phase?...Ch. 11 - Prob. 54TECh. 11 - The left most diagram below shows the moving...Ch. 11 - The leftmost diagram here shows two phases of a...Ch. 11 - A cotton ball is dipped in alcohol and wiped...Ch. 11 - A skillet is lined with a thin layer of cooking...Ch. 11 - A cotton ball is dipped in alcohol is wiped across...Ch. 11 - Use exercise 58 as an analogy to describe what...Ch. 11 - Prob. 61TECh. 11 - Prob. 62TECh. 11 - Prob. 63TECh. 11 - Why are physical changes typically easier to...Ch. 11 - Prob. 65TECh. 11 - Prob. 66TECh. 11 - Each night you measure your height just before...Ch. 11 - State whether each of the following is an example...Ch. 11 - State whether each of the following is an example...Ch. 11 - How is sugar dissolving in water an example of a...Ch. 11 - Why is the air over a campfire always moist?Ch. 11 - Prob. 72TECh. 11 - Prob. 73TECh. 11 - Each sphere in the diagrams shown here represents...Ch. 11 - Is aging primarily an example of a physical or a...Ch. 11 - Is nuclear fusion, as described in Chapter 10, an...Ch. 11 - Prob. 77TECh. 11 - Prob. 78TECh. 11 - Oxygen atoms are used to make water molecules....Ch. 11 - Oxygen, O2, is certainly good for you. Does it...Ch. 11 - Prob. 81TECh. 11 - Prob. 82TECh. 11 - Which of the following boxes contains only an...Ch. 11 - Prob. 84TECh. 11 - Prob. 85TECh. 11 - What is the chemical name for a compound with the...Ch. 11 - Prob. 87TECh. 11 - Prob. 88TECh. 11 - Is nanotechnology the result of basic or applied...Ch. 11 - How does a scanning probe microscope differ from...Ch. 11 - People often behave differently in a group...Ch. 11 - Prob. 92TECh. 11 - Medicines, such as pain relievers and...Ch. 11 - Your friend smells cinnamon coming from an...Ch. 11 - The British diplomat, physicist, and...Ch. 11 - Prob. 96TDICh. 11 - A calculator is useful but certainly not exciting....Ch. 11 - How might speculations about potential dangers of...Ch. 11 - Over the past 20 years, the average life...Ch. 11 - Prob. 100TDICh. 11 - Prob. 1RATCh. 11 - The molecules in a small collection of molecules...Ch. 11 - The phase in which atoms and molecules no longer...Ch. 11 - Prob. 4RATCh. 11 - Prob. 5RATCh. 11 - Prob. 6RATCh. 11 - Which is an example of a chemical change? a Water...Ch. 11 - If you burn 50kg of wood and produce 10g of ash,...Ch. 11 - If you have one molecule of TiO2, how many...Ch. 11 - Prob. 10RAT
Additional Science Textbook Solutions
Find more solutions based on key concepts
Choose the best answer to each of the following. Explain your reasoning. The solar nebula was 98% (a) rock and ...
Cosmic Perspective Fundamentals
If 10,000 people saw the same UFO, scientists would be forced to conclude that an alien visit really occurred.
...
Life in the Universe (4th Edition)
3.15 Inside a starship at rest on the earth, a ball rolls off the top of a horizontal table and lands a distanc...
University Physics with Modern Physics (14th Edition)
Use your knowledge of the velocities and changes in velocities to construct momentum vectors and change in mome...
Tutorials in Introductory Physics
For the particle in Problem 49, is there any time t 0 when the particle is (a) at rest and (b) accelerating in...
Essential University Physics: Volume 1 (3rd Edition)
The pV-diagram of the Carnot cycle.
Sears And Zemansky's University Physics With Modern Physics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- The volume of a sample of oxygen gas changed from 10 mL to 11 mL as the temperature changed. Is this a chemical or physical change? Why?arrow_forwardWhat is Fick’s law? How this law is modified if guest molecules in fluid are dispersed with certain velocity.arrow_forwardThe heat capacity of a solid at constant volume is given, according to the Debye model, assee figure where ΘD is the Debye temperature, N the number of atoms, and kB the Boltzmann constant. Show that this tends to a constant at high temperatures.arrow_forward
- The heat capacity of a solid at constant volume is given, according to the Debye model, as see figure where ΘD is the Debye temperature, N the number of atoms, and kB the Boltzmann constant. Show that this tends to a constant at high temperatures.arrow_forwardPlease answer question and just send me the paper solutions asap dont type the answer question 6 use the given formula only ASAParrow_forwardWhen eating a piece of hot apple pie, you may find that the crust is only warm but the apple filling burns your mouth. Why?arrow_forward
- How do you solve these questions?This is also just these questions with nothing added to it.arrow_forwardIf flat strips of iron and brass were bonded together and this bimetallic strip were heated, what would be observed ? Justify your answer, and draw a sketch of the situation. Alpha for iron is 1.14 x 10^-5 degree Celsius. Alpha for brass is 1.88 x 10^-5 degree celsiusarrow_forwardPlease Kindly Help me to answer this. Thank you very much! Note: Round off your final answer to the nearest ten-thousandth. Take note: Only the final answer should be rounded off. Do NOT write answers in scientific notation. Always write the answer with 4 decimal places, (i.e, 4.25 should be written as 4.2500) EXCEPT for whole numbers. Question: Steel railroad tracks are laid when the temperature is 5.0 °C. A standard section of rail is then 12.0 m long. The gap should be left between rail sections so that there is no compression when the temperature gets as high as 42 °C is ____________ cm.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY