
(a)
Interpretation: The reagents that can be used to achieve the following transformation are to be identified.

Concept introduction: The given compound is a terminal
(b)
Interpretation: The reagents that can be used to achieve each of the following transformations are to be identified.

Concept introduction: The starting alkyne is a compound with five carbon atoms, which to be converted into a seven carbon-containing terminal alkyne, needs to undergo a reaction with such reagents which can facilitate this reaction. Reduction using a poisoned catalyst such as Lindlar’s catalyst, followed by bromination and reaction with an alkynide can yield the desired product.
(c)
Interpretation: The reagents that can be used to achieve each of the following transformations are to be identified.

Concept introduction: The starting material has one more carbon atom than the product. This means the synthesis must have an ozonolysis process, to cleave a carbon-carbon bond. Also, since the product is a
(d)
Interpretation: The reagents that can be used to achieve each of the following transformations are to be identified.
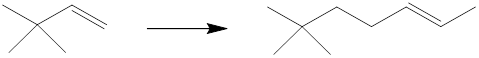
Concept introduction: The starting material has six carbon atoms, and the product has nine carbon atoms. So the synthesis must involve the installation of three carbon atoms and also, the location of the functional group has been changed. The starting material is a cis-alkene while the product is a trans-alkene. The starting material must be alkylated with an alkynide which can be reduced with a dissolving metal to yield the product.
(e)
Interpretation: The reagents that can be used to achieve each of the following transformations is to be identified.
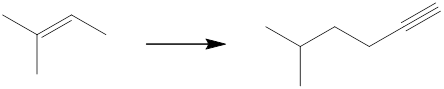
Concept introduction: The product has two more carbon atoms than the starting material, and the location of the functional group has changed. So, bromination followed by dehydrohalogenation can give a terminal alkene, which on further bromination followed by reaction with an alkynide would yield the desired product.
(f)
Interpretation: The reagents that can be used to achieve each of the following transformation is to be identified.
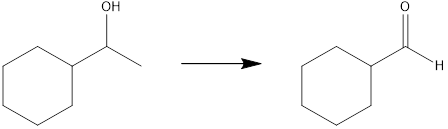
Concept introduction: The starting material has one more carbon atom than the product. Therefore, the synthesis must employ an ozonolysis process, to cleave a carbon-carbon bond. For the formation of an aldehyde product, an alkene is also required. For this alkene to be formed, the alcohol must be converted to a tosylate, and then after the alkene is formed, it can undergo ozonolysis to yield the product.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardA synthetic organic molecule, G, which contains both aldehyde and ether functional groups, is subjected to a series of reactions in a multi-step synthesis pathway. In the first step, G undergoes a Wittig reaction, leading to the formation of an alkene, H. Subsequently, H is treated with an ozone (O3) reagent followed by a reducing agent in an ozonolysis reaction, resulting in the formation of two different products, I and J. Considering the functional groups present in G and the nature of the reactions involved, what are the most probable structures or functional groups present in products I and J? A. I contains a carboxylic acid group, and J contains an aldehyde group. B. I contains a ketone group, and J contains an alcohol group. C. I and J both contain aldehyde groups. D. I contains an ester group, and J contains a ketone group. Don't use chat gpt.arrow_forwardFill in the reagents for the following transformations. NO2 NH2 NEN CI CNarrow_forward
- The following molecule belongs to a class of compounds called enediols. Each carbon of the double bond carries an —OH group: Draw structural formulas for the α-hydroxyketone and the α-hydroxyaldehyde with which this enediol is in equilibrium. α-hydroxyketone α-hydroxyaldehydearrow_forwardThe following compound undergoes an intramolecular reaction to form ethene and a product with a five-membered ring. Identify the product and the catalyst used to carry out the reaction.arrow_forwardAcid-catalyzed dehydration of 3-methyl-2-butanol gives three alkenes: 2-methyl-2- butene, 3-methyl-1-butene, and 2-methyl-1-butene. Propose a mechanism to account for the formation of each productarrow_forward
- The following molecule belongs to a class of compounds called enediols; each carbon of the double bond carries an-OH group. Draw structural formulas for the a-hydroxyketone and the a-hydroxyaldehyde with which this enediol is in equilibrium. CH-OH a-Hydroxyaldehyde = C-OH= a -Hydroxyketone ČH3 An enediolarrow_forwardName the following compound.arrow_forwardy-Butyrolactone (C4H6O2, GBL) is a biologically inactive compound that is converted to the biologically active recreational drug GHB (Section 19.5) by a lactonase enzyme in the body. Since y-butyrolactone is more fat soluble than GHB, it is more readily absorbed by tissues and thus produces a faster onset of physiological symptoms. y-Butyrolactone shows an absorption in its IR spectrum at 1770 cm-1 and the following 1H NMR spectral data: 2.28 (multiplet, 2 H), 2.48 (triplet, 2 H), and 4.35 (triplet, 2 H) ppm. What is the structure of y-butyrolactone?arrow_forward
- REACTIONS Indicate the reagent(s) needed to carry out the following transformations. a) он COTS b) онarrow_forwardThe reaction of a ketone with an alcohol in acidic conditions will result in: an alkane an acetal a geminal (gem) diol an alkenearrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


