
Concept explainers
For each compound. [1] Identify the functional group; [2] draw out the complete compound, including lone pairs on heteratoms.
![Chapter 11.5, Problem 11.8PP, For each compound. [1] Identify the functional group; [2] draw out the complete compound, including , example 1](http://dev-ingestion-image-output.s3-website-us-east-1.amazonaws.com/9781259883989/Chapter-11/images/html_83989-11.5-11.8pp_image001.jpg)
b.
![Chapter 11.5, Problem 11.8PP, For each compound. [1] Identify the functional group; [2] draw out the complete compound, including , example 2](http://dev-ingestion-image-output.s3-website-us-east-1.amazonaws.com/9781259883989/Chapter-11/images/html_83989-11.5-11.8pp_image002.jpg)
d.
![Chapter 11.5, Problem 11.8PP, For each compound. [1] Identify the functional group; [2] draw out the complete compound, including , example 3](http://dev-ingestion-image-output.s3-website-us-east-1.amazonaws.com/9781259883989/Chapter-11/images/html_83989-11.5-11.8pp_image003.jpg)
(a)
Interpretation:
The functional group / groups should be identified and the complete structure with lone pairs on hetero-atoms of the following compound should be drawn:
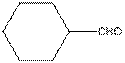
Concept Introduction:
Organic molecules have some structural features in addition to
This table gives information about the compounds containing
| Type of Compound | General Structure |
| Aldehyde | 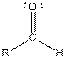 |
| Ketone | 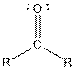 |
| Carboxylic acid | 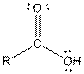 |
| Ester | 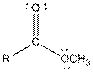 |
| Amide | 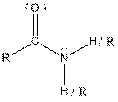 |
Here, R = any carbon backbone
Complete structural formula of a molecule represents all the atoms of molecule, types of bonds connecting atoms and how atoms are connected to each other.
Answer to Problem 11.8PP
Functional group in the given compound is '
Explanation of Solution
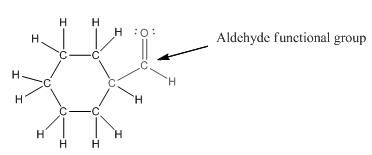
The given compound is as follows:
The given compound can also be drawn as:
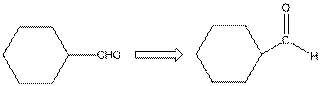
This compound contains a six membered ring and a
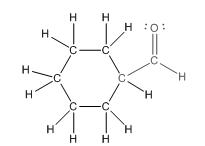
To draw the complete structure of the given skeletal structure, place carbon atom on each vertices and fulfill its valency with hydrogen atoms. Unlabelled vertex will represent carbon atom attached to number of hydrogen atoms required to fulfill its valency (i.e. four). Valence electron of oxygen is six. Out of six two electrons are used in making double bond with carbon atom. Then remaining four electrons will be available as lone pair.
So, complete structure of given compound is as follows:
(b)
Interpretation:
The functional group / groups should be identified and the complete structure with lone pairs on hetero-atoms of the following compound should be drawn:
Concept Introduction:
Organic molecules have some structural features in addition to
This table gives information about the compounds containing
| Type of Compound | General Structure |
| Aldehyde | 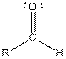 |
| Ketone | 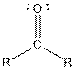 |
| Carboxylic acid | 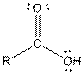 |
| Ester | 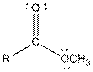 |
| Amide | 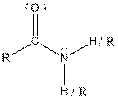 |
Here, R = any carbon backbone
Complete structural formula of a molecule represents all the atoms of molecule, types of bonds connecting atoms and how atoms are connected to each other.
Answer to Problem 11.8PP
Functional group in the given compound
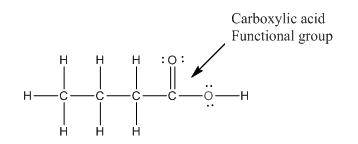
Explanation of Solution
The given compound is as follows:
The above compound can be drawn as:
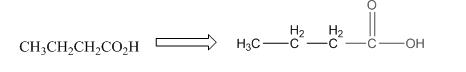
This compound contains four carbon and eight hydrogen atom with two oxygen atoms. The functional group is carboxylic acid containing an
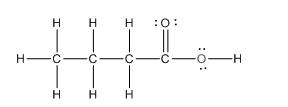
To draw the complete structure of the given skeletal structure, place carbon atom on each vertices and fulfill its valency with hydrogen atoms. Unlabelled vertex will represent carbon atom attached to number of hydrogen atoms required to fulfill its valency (i.e. four). Valence electron of oxygen is six. Out of six two electrons of blue colored oxygen are used in making double bond with carbon atom. Then remaining four electrons will be available as lone pair.
Complete structure of the given compound is as follows:
(c)
Interpretation:
The functional group / groups should be identified and the complete structure with lone pairs on hetero-atoms of the following compound should be drawn:
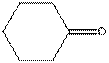
Concept Introduction:
Organic molecules have some structural features in addition to
This table gives information about the compounds containing
| Type of Compound | General Structure |
| Aldehyde | 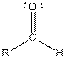 |
| Ketone | 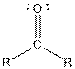 |
| Carboxylic acid | 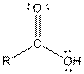 |
| Ester | 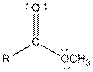 |
| Amide | 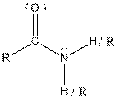 |
Here, R = any carbon backbone
Complete structural formula of a molecule represents all the atoms of molecule, types of bonds connecting atoms and how atoms are connected to each other.
Answer to Problem 11.8PP
Functional group in the given compound is 'ketone' and the complete structure of the compound is as follows:
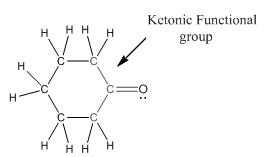
Explanation of Solution
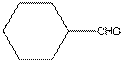
The given compound is as follows:
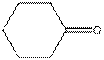
This compound contains six membered ring with one oxygen atom. The functional group is ketone containing a carbon-oxygen bond
To draw the complete structure of the given skeletal structure, place carbon atom on each vertices and fulfill its valency with hydrogen atoms. Unlabelled vertex will represent carbon atom attached to number of hydrogen atoms required to fulfill its valency (i.e. four). Valence electron of oxygen is six. Out of six two electrons of are used in making double bond with carbon atom. Then remaining four electrons will be available as lone pair.
Complete structure of the compound is as follows:
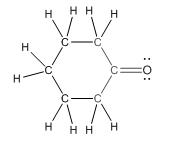
(d)
Interpretation:
The functional group / groups should be identified and the complete structure with lone pairs on hetero-atoms of the following compound should be drawn:
Concept Introduction:
Organic molecules have some structural features in addition to
This table gives information about the compounds containing
| Type of Compound | General Structure |
| Aldehyde | 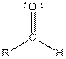 |
| Ketone | 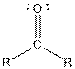 |
| Carboxylic acid | 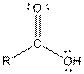 |
| Ester | 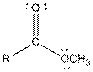 |
| Amide | 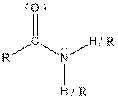 |
Here, R = any carbon backbone
Complete structural formula of a molecule represents all the atoms of molecule, types of bonds connecting atoms and how atoms are connected to each other.
Answer to Problem 11.8PP
Functional group in the given compound is 'ester' and the complete structure of the compound is as follows:
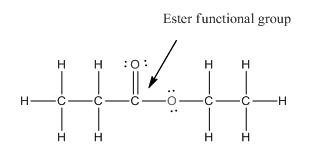
Explanation of Solution
The given compound is as follows:
This given compound can be drawn as follows:
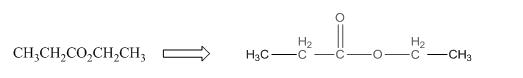
This compound contains an
To draw the complete structure of the given skeletal structure, place carbon atom on each vertices and fulfill its valency with hydrogen atoms. Unlabelled vertex will represent carbon atom attached to number of hydrogen atoms required to fulfill its valency (i.e. four).
Valence electron of oxygen is six. Out of six two electrons of blue colored oxygen are used in making double bond with carbon atom. Then remaining four electrons will be available as lone pair. For pink colored oxygen atom, one electron is used in making bond with carbonyl carbon atom and another electron is used in making bond with carbon backbone. Then remaining four will be present as lone pair.
The complete structure of the given compound is as follows:
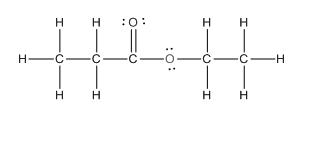
(e)
Interpretation:
The functional group / groups should be identified and the complete structure with lone pairs on hetero-atoms of the following compound should be drawn:
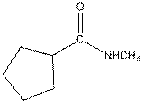
Concept Introduction:
Organic molecules have some structural features in addition to
This table gives information about the compounds containing
| Type of Compound | General Structure |
| Aldehyde | 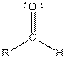 |
| Ketone | 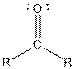 |
| Carboxylic acid | 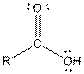 |
| Ester | 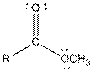 |
| Amide | 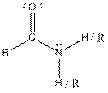 |
Here, R = any carbon backbone
Complete structural formula of a molecule represents all the atoms of molecule, types of bonds connecting atoms and how atoms are connected to each other.
Answer to Problem 11.8PP
Functional group in the given compound is an 'amide' and the complete structure of the compound is as follows:
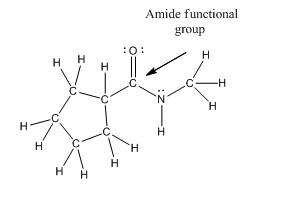
Explanation of Solution
The given compound is as follows:
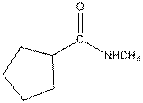
This compound contains an
To draw the complete structure of the given skeletal structure, place carbon atom on each vertices and fulfill its valency with hydrogen atoms. Unlabelled vertex will represent carbon atom attached to number of hydrogen atoms required to fulfill its valency (i.e. four).
Valence electron of oxygen is six. Out of six two electrons of oxygen atom are used in making double bond with carbon atom. Then remaining four electrons will be available as lone pair. Valence electron of nitrogen is five. Out of five, one electron is used in making bond with the carbonyl carbon atom, one electron is used in making bond with
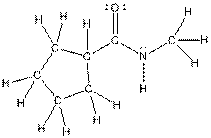
The complete structure of the given compound is as follows:
Want to see more full solutions like this?
Chapter 11 Solutions
GEN ORG + BIO (LL) W/CONNECT
- Identify each compound according to its functional group:arrow_forwardConvert each condensed formula to a Lewis structure. CH3(CH2)4CH(CH3)2 (CH3)3CCH(OH)CH2CH3 (CH3)2CHCHO (HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardHi! Could someone help me find the molecular formula for this structure? I can’t figure it out and I’m slightly confused. Thank you so much!arrow_forward
- Convert each condensed formula to a Lewis structure. a.CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c. (CH3)2CHCHO d.(HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardDraw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH3CH2)2O, the rst general anesthetic used in medical proceduresb. acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon bersc. dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning productsd. acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forwardMake 2 isomers out of C4H9O, Include a drawing and the namesarrow_forward
- 3. a. What is the chemical structure of benzoic acid, circle functional groups different than alkane,alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? __________________________arrow_forwardConvert each shorthand structure to a complete structure with all atoms and lone pairs drawn in. (CH3)3COH =arrow_forwardCan you help me with parts d,e,and f for the compound (ch3)2cchco2harrow_forward
- Each alkane formula listed below has an error in it. Identify and state the error, then rewrite the formula correctly with the same number of carbon atoms. CH5 C2H3 C6H7 CH2Oarrow_forwardthe context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (h).arrow_forwardConvert each condensed formula to a complete structure with lone pairs on heteroatoms. a. CH 3(CH 2) 8CH 3 c. CH 3CCl 3 e. (CH 3) 2CHCH 2NH 2 b. CH 3(CH 2) 4OH d. CH 3(CH 2) 4CH(CH 3) 2arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


