
Concept explainers
What products would you expect from the reaction of 1-bromopropane with each of the following?
(a) NaNH2
(b) KOC(CH3)3
(c) NaI
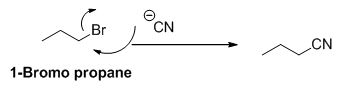
(d) NaCN
(e) NaC≡CH
(f) Mg, then H2O
a)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
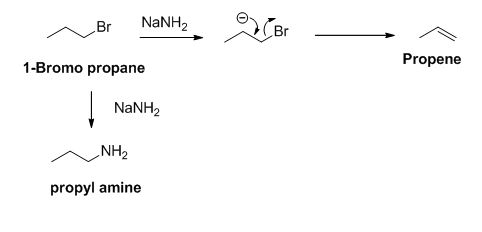
Explanation of Solution
Given information:
The reactant of the reaction is given below,
The product of the reaction is given below,
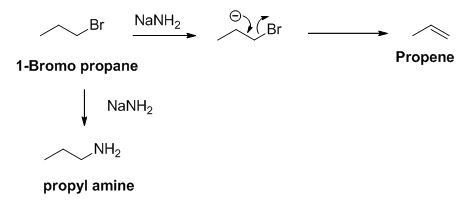
The sodium amide is acts as a base and it abstract the acidic proton from 1-bromo propane gives carbanion, this carbanion under goes elimination reaction gives propene as a major product.
1-bromo propane undergoes nucleophilic substitution reaction gives propyl amine as a minor product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
b)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
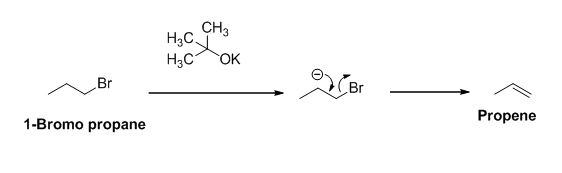
Explanation of Solution
Given information:
The reactant of the reaction is given below,
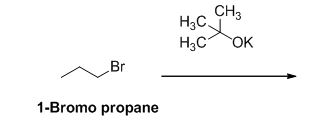
The product of the reaction is given below,
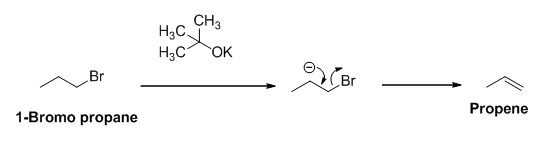
The potassium tertiary butoxide is acts as a base and it abstract the acidic proton from 1-bromo propane gives carbanion, this carbanion under goes elimination reaction gives propene as a major product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
c)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
Explanation of Solution
Given information:
The reactant of the reaction is given below,
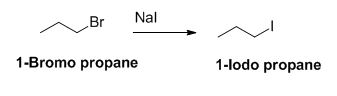
The product of the reaction is given below,
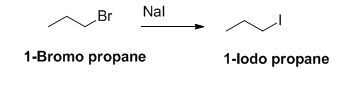
The sodium iodide is acts as nucleophile, 1-bromo propane undergoes nucleophilic substitution reaction with sodium iodide gives 1-iodo propane.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
d)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
Explanation of Solution
Given information:
The reactant of the reaction is given below,
The product of the reaction is given below,
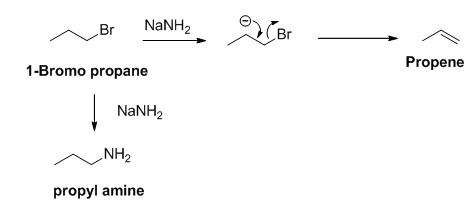
The sodium amide is acts as a base and it abstract the acidic proton from 1-bromo propane gives carbanion, this carbanion under goes elimination reaction gives propene as a major product.
1-bromo propane undergoes nucleophilic substitution reaction gives propyl amine as a minor product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
e)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
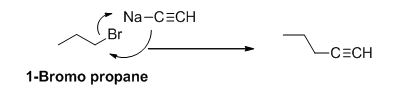
Explanation of Solution
Given information:
The reactant of the reaction is given below,
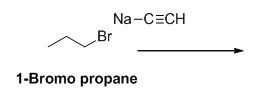
The product of the reaction is given below,
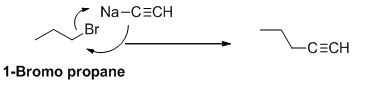
1-bromo propane undergoes nucleophilic substitution reaction with given alkyne compound gives the corresponding alkyne product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
f)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
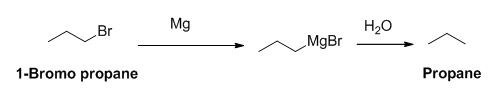
Explanation of Solution
Given information:
The reactant of the reaction is given below,
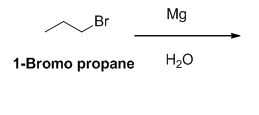
The product of the reaction is given below,
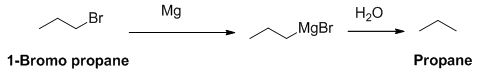
1-bromo propane reaction with magnesium gives Grignard reagent and it undergoes hydrolysis gives propane.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry - Owlv2 Access (4 Term)
- The following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardWhen 5-bromo-1-pentanol is treated with sodium hydride in diethyl ether, the product is analyzed to be C5H10O. Propose a likely structure for this product, suggesting a reasonable mechanistic pathway for its formationarrow_forwardz When (R)-2-butanol is left standing in aqueous acid, it slowly loses its optical activity. Account for this observationarrow_forward
- An allylic halide with the chemical formula C5H9Cl undergoes an SN1 reaction with water to produce B and C, two isomeric products. What would be the structure of the allylic halide and the products B and C? What peaks correspond with what protons in B and C?arrow_forwardTreatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forwardWhen 2-methyl-2, 5-pentanediol is treated with sulfuric acid, dehydration occurs and 2, 2-dimethyltetrahydrofuran is formed. Suggest a mechanism for this reaction. Which of the two oxygen atoms is most likely to be eliminated, and why?arrow_forward
- When 2,2-dibromo-1-phenylpropane is heated overnight with sodium amide at 150 °C, the major product (after addition of water) is a different foul-smelling compound of formula C9H8. Propose a structure for this product, and give a mechanism to account for its formation.arrow_forwardThe UV spectrum of 1-phenylprop-2-en-1-ol shows an intense absorption at 220 nm and a weaker absorption at 258 nm. When this compound is treated with dilute sulfuric acid, it rearranges to an isomer with an intense absorption at 250 nm and a weaker absorption at 290 nm. Suggest a structure for the isomeric product and propose a mechanism for its formation.arrow_forwardThe treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formulaC5H10O) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.arrow_forward
- A. In the synthesis of 1-bromobutane, what is the inorganic by-product left in the reaction flask following the distillation? Why was the bromoalkane the bottom layer in the separatory funnel? B. Predict the product when 1-methylcyclohexanol reacts with H2SO4 and KBr. Show the mechanism.arrow_forward3a)Give the mechanisms for the following transformations:arrow_forwardWhen 2,2-dibromo-1-phenylpropane is heated overnight in fused KOH at 200 °C, the major product is a foul-smelling compound of formula C9H8. Propose a structure for this product, and give a mechanism to account for its formation.arrow_forward

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


