
Concept explainers
Interpretation:
The following compounds are to be matched with their respective IR spectra.
(a)
(b)
(c)
(d) dipropyl ether
(e)
(f) cyclohexane
(g)
Concept introduction:
The IR spectrum of a compound shows absorptions at different frequencies. These absorption frequencies depend upon the bond stretching frequency of the bonds in molecule.
Answer to Problem 12.27AP
The matching of the given compounds with their respective IR spectra is shown in the table below.
| Compounds | IR spectra |
| a. |
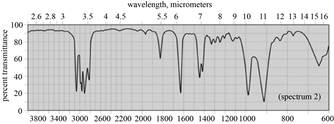 |
| b. |
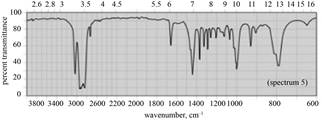 |
| c. |
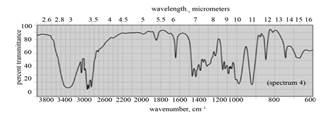 |
| d. dipropyl ether | 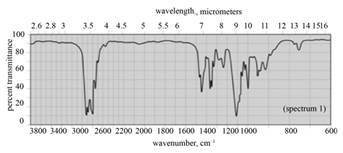 |
| e. |
No IR spectrum available. |
| f. cyclohexane | 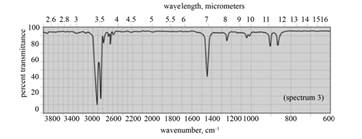 |
| g. |
No IR spectrum available. |
Explanation of Solution
(a) The given compound is
Figure 1
In the IR spectra of
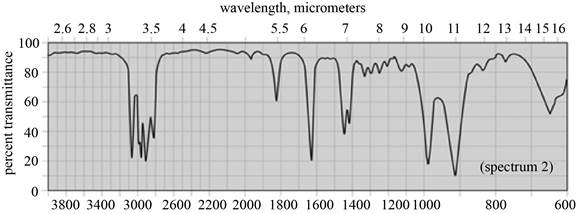
Figure 2
(b) The given compound is
Figure 3
In the IR spectra of
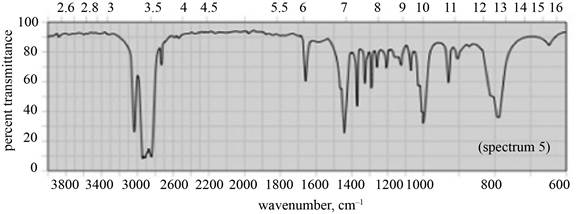
Figure 4
(c) The given compound is
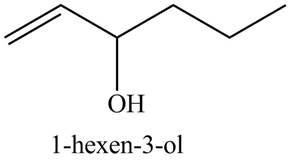
Figure 5
In the IR spectra of
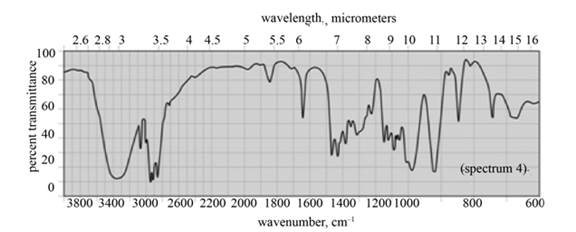
Figure 6
(d) The given compound is dipropyl ether,
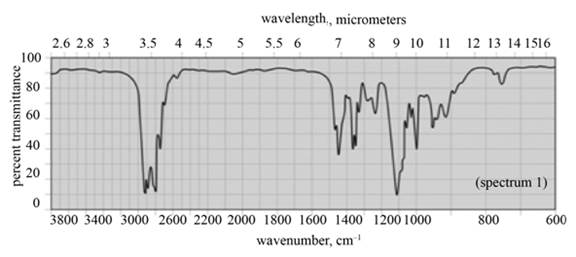
Figure 7
(e) The IR spectrum for
(f) The given compound is cyclohexane as shown below.
Figure 8
In the IR spectra of cyclohexane, a strong absorption peak for
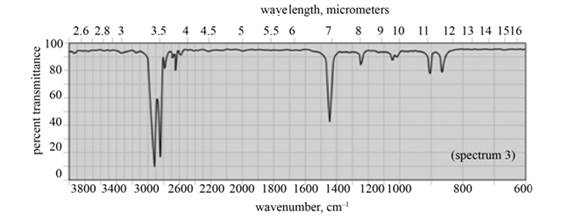
Figure 9
(g) The IR spectrum for
(a) The spectrum 2 corresponds to the compound,
(b) The spectrum 5 corresponds to the compound,
(c) The spectrum 4 corresponds to the compound,
(d) The spectrum 1 corresponds to the compound, dipropyl ether.
(e) There is no spectrum available for the compound,
(f) The spectrum 3 corresponds to the compound, cyclohexane.
(g) There is no spectrum available for the compound,
Want to see more full solutions like this?
Chapter 12 Solutions
ORGANIC CHEMISTRY (LL)+ SAPLING ACC >BI
- Part 3B Set 1. Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. Predict the 1H NMR spectrum for each compound (include integration, multiplicity, and approximate chemical shift). Put it in data table format.arrow_forwardBased on the carbon NMR spectrum attached below, how many carbons are present in your unknown and does this agree with the proposition that this NMR was of C6H12O2? Explain.arrow_forwardWhich molecule is consistent with both of these IR and 1H NMR spectra?arrow_forward
- A Grignard reaction was conducted using bromobenzene and benzaldehyde to make diphenylmethanol. How can we know that the IR spectra for diphenylmethanol is in fact diphenylmethanol apart from the title, keeping in mind the spectra of the reactants? Mention specific wavenumber and their corresponding functional groups.arrow_forwardWhat bond(s) do alkenes and aromatic rings have in common? Can their peaks be differentiated using IR spectroscopy? What about the C-H bonds in alkanes and alkenes, can they be differentiated using IR spectroscopy? If so, briefly explain how. If not, explain why not.arrow_forwardWHich of these molecules best corresponds to IR spectrum shown. and why?arrow_forward
- 1. Predict the maximum total number of possible peaks in the 13C NMR spectrum of sesamin (Hint: , keep in mind the molecule’s symmetry when predicting the # of NMR peaks) a. Indicate the number of non-aromatic peaks in the 13C NMR of sesamin b.Indicate the number of aromatic peaks in the 13C NMR of sesaminarrow_forwardWhat information can be obtained from the UV-visible spectrum for compound 1 and 2?arrow_forwardThe 1HNMR signals the alkene protons of three ethylene derivatives with different substitution patterns are shown below. Considering that each of the derivatives contains AT MOST two substituents (X and/or Y) and at least two protons. A) Propose the possible substitution patterns for each of the derivatives based on the observed splitting patterns. Justify your proposals B) How might one differentiate between the three possible isomers of derivative 3 in the question above using 1H NMR spectroscopy? Explain in a sentence – no calculation is needed. C) Give TWO methods that can be used to differentiate between the three possible isomers of derivative 2 in the question above using 1H NMR spectroscopy? Explain in a sentence – no calculation is needed.arrow_forward
- Which of the following is expected to show 3 signals in the 1H NMR? (please explain in simple terms please)arrow_forward(a) The 'H-NMR spectrum of cyclobutanone shows two signals - signal A at 3.00 ppm and signal B at 1.95 ppm. Give the multiplicity of each signal. cyclobutanone (b) When cyclobutanone is treated with D20 and NaOD, the only signal observable in the 1H-NMR is a singlet at 2.00 ppm. Explain why this is the case. [Note: Deuterium atoms do not display signals in the TH-NMR spectrum]arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

