
Package: Organic Chemistry With Connect 2-semester Access Card
4th Edition
ISBN: 9781259671838
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.69P
Devise a synthesis of each compound from the indicated starting material and any other required reagents.
a.
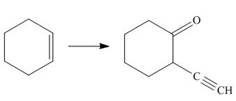
b. 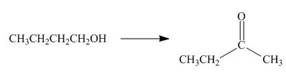 d.
d. 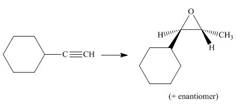
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Devise a synthesis of each substituted cyclopropane. Use acetylene (HC=CH) as a starting material in part (a) and cyclohexanone as a starting material in part (b). You may use any other organic compounds and any needed reagents.
Devise a synthesis of each compound from the indicated starting material:
Devise a synthesis of each alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents.
Chapter 12 Solutions
Package: Organic Chemistry With Connect 2-semester Access Card
Ch. 12 - Prob. 12.1PCh. 12 - Prob. 12.2PCh. 12 - Prob. 12.3PCh. 12 - Prob. 12.4PCh. 12 - Prob. 12.5PCh. 12 - Given that syn addition of H2 occurs from both...Ch. 12 - Compound Molecular formula before...Ch. 12 - Draw the products formed when triacylglycerol A is...Ch. 12 - Prob. 12.9PCh. 12 - Prob. 12.10P
Ch. 12 - Problem 12.11 (a) Draw the structure of a compound...Ch. 12 - Prob. 12.12PCh. 12 - Prob. 12.13PCh. 12 - Prob. 12.14PCh. 12 - Prob. 12.15PCh. 12 - Prob. 12.16PCh. 12 - Prob. 12.17PCh. 12 - Problem 12.18 Draw the products formed when both...Ch. 12 - Prob. 12.19PCh. 12 - Prob. 12.20PCh. 12 - Prob. 12.21PCh. 12 - Prob. 12.22PCh. 12 - Prob. 12.23PCh. 12 - Problem 12.24 Draw the organic products in each of...Ch. 12 - Prob. 12.25PCh. 12 - Prob. 12.26PCh. 12 - Problem 12.27 Draw the products of each Sharpless...Ch. 12 - Prob. 12.28PCh. 12 - 12.29 Draw the products formed when A is treated...Ch. 12 - Prob. 12.30PCh. 12 - 12.31 Devise a synthesis of the following compound...Ch. 12 - Label each reaction as oxidation, reduction, or...Ch. 12 - Prob. 12.33PCh. 12 - Prob. 12.34PCh. 12 - Prob. 12.35PCh. 12 - Prob. 12.36PCh. 12 - 12.37 Stearidonic acid (C18H28O2) is an...Ch. 12 - Draw the organic products formed when cyclopentene...Ch. 12 - Prob. 12.39PCh. 12 - Draw the organic products formed when allylic...Ch. 12 - Draw the organic products formed in each reaction...Ch. 12 - Draw the organic products formed in each reaction....Ch. 12 - Prob. 12.43PCh. 12 - Prob. 12.44PCh. 12 - Prob. 12.45PCh. 12 - What alkene is needed to synthesize each 1,2-diol...Ch. 12 - Prob. 12.47PCh. 12 - Draw the products formed after Steps 1 and 2 in...Ch. 12 - Prob. 12.49PCh. 12 - Prob. 12.50PCh. 12 - Prob. 12.51PCh. 12 - What alkyne gives each set of products after...Ch. 12 - Prob. 12.53PCh. 12 - Prob. 12.54PCh. 12 - Prob. 12.55PCh. 12 - 12.54 An unknown compound A of molecular formula ...Ch. 12 - 12.55 DHA is a fatty acid derived from fish oil...Ch. 12 - Prob. 12.58PCh. 12 - Prob. 12.59PCh. 12 - 12.58 Epoxidation of the following allylic alcohol...Ch. 12 - What allylic alcohol and DET isomer are needed to...Ch. 12 - Devise a synthesis of each hydrocarbon from...Ch. 12 - Prob. 12.63PCh. 12 - 12.62 It is sometimes necessary to isomerize a cis...Ch. 12 - Prob. 12.65PCh. 12 - Prob. 12.66PCh. 12 - Prob. 12.67PCh. 12 - Prob. 12.68PCh. 12 - Devise a synthesis of each compound from the...Ch. 12 - Devise a synthesis of each compound from acetylene...Ch. 12 - Prob. 12.71PCh. 12 - Prob. 12.72PCh. 12 - Prob. 12.73PCh. 12 - Prob. 12.74PCh. 12 - Prob. 12.75PCh. 12 - Prob. 12.76PCh. 12 - 12.72 Draw a stepwise mechanism for the following...Ch. 12 - Prob. 12.78PCh. 12 - Prob. 12.79PCh. 12 - Prob. 12.80PCh. 12 - 12.75 Sharpless epoxidation of allylic alcohol X...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the appropriate reagent over each arrow.arrow_forwardDevise a synthesis of each compound using a Heck reaction as one step. You may use benzene, CH2=CHCO2Et, organic alcohols having one or two carbons, and any required inorganic reagents.arrow_forwardShow how you might use the Williamson ether synthesis to prepare each ether. (a) (b)arrow_forward
- How Wittig reagents are synthesized by a two-step procedure ?arrow_forwardDraw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.arrow_forwardWhat product is formed when acetic acid is treated with each reagent: (a) CH3NH2; (b) CH3NH2, then heat; (c) CH3NH2 + DCC?arrow_forward
- Draw the product formed when (CH3)2CHOH is treated with each reagent (d, e and f)arrow_forwardDevise a synthesis of each compound using 1-bromobutane(CH3CH2CH2CH2Br) as the only organic starting material. You may useany other inorganic reagents.arrow_forwardDevise a synthesis of attached alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents.arrow_forward
- Whats the reason for the girgnard reagent attack that carbon of starting point in this step? a. Bc it gives us desired product b. Due to the sterics c. Bc it is more electrophilic than the other carbon of the starting pointarrow_forwardβ-D-Glucose, a hemiacetal, can be converted to a mixture of acetals on treatment with CH3OH in the presence of acid. Draw a stepwise mechanism for this reaction. Explain why two acetals are formed from a single starting material.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY