
Solid objects, such as your desk or a rod of aluminum, can conduct heat. The magnitude of the thermal diffusivity of the material determines how quickly the heat moves through a given amount of material. The equation for thermal diffusivity (a) is given by:
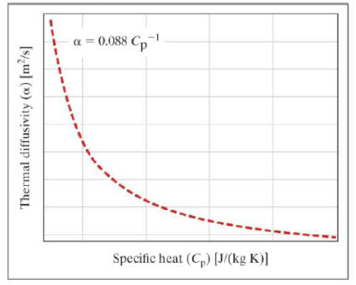 Experiments are conducted to change the specific heat (Cp) of the material while holding the thermal conductivity (k) and the density (ρ) constant. The results are shown graphically.
Experiments are conducted to change the specific heat (Cp) of the material while holding the thermal conductivity (k) and the density (ρ) constant. The results are shown graphically.
- a. What are the units of the constant 0.088? Simplify your answer.
- b. If the thermal conductivity of the material is 237 watts per meter kelvin, what is the density of the material?
- c. If the material has a density of 4500 kilograms per cubic meter, what is the thermal conductivity of the material in units of watts per meter kelvin?
Learn your wayIncludes step-by-step video

Chapter 12 Solutions
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
Additional Engineering Textbook Solutions
Engineering Mechanics: Statics
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Engineering Mechanics: Dynamics (14th Edition)
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
Statics and Mechanics of Materials (5th Edition)
- 1.77 Explain each in your own words. (a) What is the mode of heat transfer through a large steel plate that has its surfaces at specified temperatures? (b) What are the modes when the temperature on one surface of the steel plate is not specified, but the surface is exposed to a fluid at a specified temperature?arrow_forwardTo determine the thermal conductivity of a structural material, a large 15-cm-thick slab of the material is subjected to a uniform heat flux of 2500 W/m2 while thermocouples embedded in the wall at 2.5 cm. intervals are read over a period of time. After the system had reached equilibrium, an operator recorded the thermocouple readings shown below for two different environmental conditions: Distance from the Surface (cm) Temperature (C) Test 1 0 40 5 65 10 97 15 132 Test 2 0 95 5 130 10 168 15 208 From these data, determine an approximate expression for the thermal conductivity as a function of temperature between 40 and 208C.arrow_forwardWhen the sun is high in the sky, it delivers approximately 1000 watts of power to each square meter of earth's surface. The temperature of the surface of the sun is about 6000 K, while that of the earth is about 300 K. Suppose you plant grass on this square meter of earth. Some people might argue that the growth of the grass (or of any other living thing) violates the second law of thermodynamics, because disorderly nutrients are converted into an orderly life form. How would you respond?arrow_forward
- An electric heater producing 260 W of heat is used to warm up a room containing 7 m3 of air. If we assume the room is perfectly sealed and there is no heat loss through the room boundaries, such that all of the heater output goes into increasing the air temperature, how long will it take to heat up the air in the room from 5.0 °C to 24.1 °C? Give your answer to the nearest minute and assume that the specific volume (v = 0.85 m3/kg) and specific heat capacity at constant volume (cv = 1.005 kJ/(kg K)) remain constant throughout the heating process.arrow_forwardLiquid is held into two components Separated by a membrane.the total volume is 775ft3 and the volume of compartment Y is 300ft3 and it’s specific volume is 9.55ft3/lb.the membrane breaks and the resulting specific volume is 12.5 ft3/lb. a) determine the original specific volume in compartment X b)after breaking the membrane,the temperature of the liquid is 3 degree centigrade.if the absolute temperature of this reading is doubled and decreased by 239 degrees,what would be the final temperature of the maximum degree Celsius and degree Rankinearrow_forwardThe block in the drawing has dimensions L0×2L0×3L0,where L0 =0.5 m. The block has a thermal conductivity of 300 J/(s·m·C˚). In drawings A, B, and C, heat is conducted through the block in three different directions; in each case the temperature of the warmer surface is 36 ˚C and that of the cooler surface is 6 ˚C Determine the heat that flows in 6 s for each case.arrow_forward
- A metal spoon and a wooden spoon are left overnight in a room. Why do we feel colder when we touch the metal spoon than when we touch the wooden one? a. Since metals usually have a lower heat capacity than non-metals, the metal spoon loose more heat to the room and consequently has a lower temperature b. Metals release heat faster than non-metals because of their higher thermal conductivity c. This is merely psychological since we are conditioned to think of metals as “cold” d. Metals absorb heat faster than non-metals because of their higher thermal conductivityarrow_forwardNewton's Law of Cooling - Differential Equations The rate at which a body cools is proportional to the difference in temperature between the body and its surroundings. If a body in air at 0°C will cool from 200°C to 100°C in 40 minutes, how many more minutes will it take the body to cool from 100°C to 50°C?arrow_forwardDE: Newton's Law of Cooling The rate at which a body cools is proportional to the difference in temperature between the body and its surroundings. If a body in air at 0°C will cool from 200°C to 100°C in 40 minutes, how many more minutes will it take the body to cool from 100°C to 50°C?arrow_forward
- Material A and material B was exposed to the same change in temperature, but the heat involved with A is 3 times greater than B. If mass of A is 1 kg and mass of B is 10 kg. how do you describe their specific heat ? a. Specific heat of A is 30 times greater compared to B b. Specific heat of A is 10 times greater compared to B d. Specific heat of A is 20 times greater compared to B c. Specific heat of A is 10 times lesser compared to Barrow_forwardLiquid nitrogen has a density of 0.808 g/mL and boils at 77 K. Researchers often purchase liquid nitrogen in insulated 175-L tanks. The liquid vaporizes quickly to gaseous nitrogen (which has a density of 1.15 g/L at room temperature and atmospheric pressure) when the liquid is removed from the tank. Suppose that all 175 L of liquid nitrogen in a tank accidentally vaporized in a lab that measured 10.00m x 10.00m x 2.50m. What maximum fraction of the air in the room could be displaced by the gaseous nitrogen?arrow_forwardQ1: If a calorimeter is not assumed to be adiabatic, how would one calculate the heat transfer? Guide Questions: What are the parameters to be considered and the equations to be used? What law of thermodynamics does this transfer of heat to the surroundings touch? Is an adiabatic system automatically considered as a closed system? Please help me understand by also attaching a reference/book/journal article. This would help me in my essay. Thank you.arrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
