
The following data are for the system
(a) How long does it take the system to reach equilibrium?
(b) How does the rate of the forward reaction compare with the rate of the reverse reaction after 30 s? After 90 s?
(a)
Interpretation:
The time required for the system to reach at equilibrium needs to be determined.
Concept introduction:
The system is said to be in equilibrium if the there is no change in the partial pressure or concentration of reactant and product takes place.
Answer to Problem 1QAP
80 s.
Explanation of Solution
The given reaction is as follows:
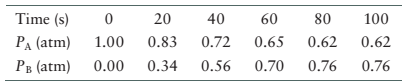
The data for the system is given as follows:
| Time (s) | |
|
| 0 | 1 | 0.000 |
| 20 | 0.83 | 0.34 |
| 40 | 0.72 | 0.56 |
| 60 | 0.65 | 0.70 |
| 80 | 0.62 | 0.76 |
| 100 | 0.62 | 0.76 |
In a system, equilibrium is a stage when the partial pressure or concentration of both reactant and product become constant or no further change in the partial pressure or concentration of reactant and as well as product takes place.
From the data, the partial pressure of reactant, A decreases from 0 s to 80 s and then become constant. In the same way, the partial pressure of product, B increases from 0 s to 90 s and then become constant. Therefore, at 80 s the concentration of both reactant and product become constant therefore, system will reach equilibrium at 80 s.
(b)
Interpretation:
The relation between the rate of forward reaction and that of the reverse reaction needs to determine after 30 s and after 90 s.
Concept introduction:
The rate of the forward reaction depends on the partial pressure of reactant and that of the reverse reaction depends on the partial pressure of the product. At equilibrium, the rate of forward reaction is equal to the rate of reverse reaction.
Answer to Problem 1QAP
After 30 s, the rate of the forward reaction is greater than the rate of the reverse reaction and after 90 s, the rate of the forward reaction is equal to the rate of the reverse reaction.
Explanation of Solution
The given reaction is as follows:
The rate expression for the given system is represented as follows:
This is the rate of the forward reaction.
The same expression for the reverse reaction will be:
At equilibrium, the rate of forward reaction is equal to the rate of backward reaction thus,
Also, at equilibrium the partial pressures of reactant and product are 0.62 atm and 0.76 atm respectively, putting the values,
Thus,
Or,
Thus, the expression for the reverse reaction will be:
Now, after 30 s the partial pressure of gas A is 0.72 atm thus, rate of forward reaction will be:
The partial pressure of gas B after 30 s is 0.56 atm thus, the rate of reverse reaction will be:
Dividing equation (1) and (2),
Thus, the rate of forward reaction is 2.12 times the rate of reverse reaction or rate of forward reaction is greater than that of the reverse reaction after 30 s.
Now, after 90 s the partial pressure of gas A is 0.62 atm thus, rate of forward reaction will be:
The partial pressure of gas B after 90 s is 0.76 atm thus, the rate of reverse reaction will be:
Dividing equation (1) and (2),
Thus, the rate of forward reaction is approximately equals to the reverse reaction after 90 s.
Want to see more full solutions like this?
Chapter 12 Solutions
Student Solutions Manual For Masterton/hurley's Chemistry: Principles And Reactions, 8th
- For the reaction N2(g)+3H2(g)2NH3(g) show that Kc = Kp(RT)2 Do not use the formula Kp = Kc(RT)5n given in the text. Start from the fact that Pi = [i]RT, where Pi is the partial pressure of substance i and [i] is its molar concentration. Substitute into Kc.arrow_forwardShow that the complete chemical equation, the total ionic equation, and the net ionic equation for the reaction represented by the equation KI(aq)+I2(aq)KI3(aq) give the same expression for the reaction quotient. KI3 is composed of the ions K+ and I3-.arrow_forwardCyclohexane, C6H12, a hydrocarbon, can isomerize or change into methylcyclopentane, a compound of the same formula (C5H9CH3) but with a different molecular structure. sssss The equilibrium constant has been estimated to be 0.12 at 25 C. If you had originally placed 0.045 mol of cyclohexane in a 2.8-L flask, what would be the concentrations of cyclohexane and methylcyclopentane when equilibrium is established?arrow_forward
- Suppose a reaction has the equilibrium constant K = 1.3 108. What does the magnitude of this constant tell you about the relative concentrations of products and reactants that will be present once equilibrium is reached? Is this reaction likely to be a good source of the products?arrow_forwardAt a certain temperature, K=0.29 for the decomposition of two moles of iodine trichloride, ICl3(s), to chlorine and iodine gases. The partial pressure of chlorine gas at equilibrium is three times that of iodine gas. What are the partial pressures of iodine and chlorine at equilibrium?arrow_forwardConsider the equilibrium N2(g)+O2(g)2NO(g) At 2300 K the equilibrium constant Kc = 1.7 103. If 0.15 mol NO(g) is placed into an empty, sealed 10.0-L flask and heated to 2300 K, calculate the equilibrium concentrations of all three substances at this temperature.arrow_forward
- For the reactionH2(g)+I2(g)2HI(g), consider two possibilities: (a) you mix 0.5 mole of each reactant. allow the system to come to equilibrium, and then add another mole of H2 and allow the system to reach equilibrium again. or (b) you mix 1.5 moles of H2 and 0.5 mole of I2 and allow the system to reach equilibrium. Will the final equilibrium mixture be different for the two procedures? Explain.arrow_forwardThe equilibrium constant Kc for the synthesis of methanol, CH3OH. CO(g)+2H2(g)CH3OH(g) is 4.3 at 250C and 1.8 at 275C. Is this reaction endothermic or exothermic?arrow_forwardWrite equilibrium-constant expressions Kp for each of the following reactions: a H2(g)+Br2(g)2HBr(g) b CS2(g)+4H2(g)CH4(g)+2H2S(g) c 4HCl(g)+O2(g)2H2O(g)+2Cl2(g) d CO(g)+2H2(g)CH3OH(g)arrow_forward
- The decomposition of PCl5(g) to form PCl3(g) and Cl2(g) has Kc = 33.3 at a high temperature. If the initial concentration of PCl5 is 0.1000 M, what are the equilibrium concentrations of the reactants and products?arrow_forwardWrite an equation for an equilibrium system that would lead to the following expressions (ac) for K. (a) K=(Pco)2 (PH2)5(PC2H6)(PH2O)2 (b) K=(PNH3)4 (PO2)5(PNO)4 (PH2O)6 (c) K=[ ClO3 ]2 [ Mn2+ ]2(Pcl2)[ MNO4 ]2 [ H+ ]4 ; liquid water is a productarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





