
Concept explainers
(a)
Interpretation:
Using necessary organic or inorganic reagents, the synthesis of given compounds has to be indicated.
Concept introduction:
Anti-Markovnikov’s rule: Unsymmetrical
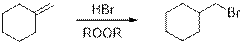
(b)
Using necessary organic or inorganic reagents, the synthesis of given compounds has to be indicated.
Concept introduction:
Anti-Markovnikov’s rule: Unsymmetrical alkene reacts with hydrogen halide, halide ions goes to the less substitution position of carbon-carbon double bond which provides alkyl halides.
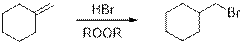
(c)
Interpretation:
Using necessary organic or inorganic reagents, the synthesis of given compounds has to be indicated.
Concept introduction:
Anti-Markovnikov’s rule: Unsymmetrical alkene reacts with hydrogen halide, halide ions goes to the less substitution position of carbon-carbon double bond which provides alkyl halides.
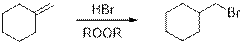
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid and gives alkene.
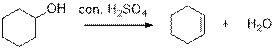
The stability of carbocation is given below,
Tertiary carbocation is more stable than the secondary and primary.
(d)
Interpretation:
Using necessary organic or inorganic reagents, the synthesis of given compounds has to be indicated.
Concept introduction:
Alkenes undergoes bromination reaction using bromine which yields the dibromo compound, bromination takes place in the double bond of the alkenes.
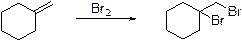
Heck reaction:
The Heck reaction is a cross-coupling reaction of an alkyl halide with an alkene to make a substituted alkenes using palladium as a catalyst and a base.
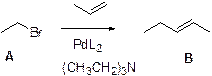
Hydrogenation:
Alkenes undergoes hydrogenation using Pd/C and hydrogen will reduce alkene to
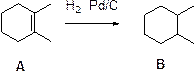
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Organic Chemistry, Books a la Carte Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- Using the given starting material, any necessary inorganic reagents and catalysts, and any carbon-containing compounds with no more than three carbons, indicate how each of the following compounds can be prepared:arrow_forwardDescribe a synthesis for each of the following compounds, using the given starting material and any necessary reagents:arrow_forwardWhat reagents should be used to prepare the following compoundsarrow_forward
- Show how the following compounds can be prepared from the given starting materials. You can use any necessary organic or inorganic reagentsarrow_forwardUsing the given starting material, any necessary inorganic reagents, and any carbon-containing compounds with no more than two carbons, indicate how each of the following compounds can be prepared:arrow_forwardStarting with cyclohexanone, show how to prepare these compounds. In addition to the given starting material, use any other organic or inorganic reagents as necessary. Q.) 1-Methylcyclohexenearrow_forward
