
Chemical/Bio Engineering
Figure P12.6 shows three reactors linked by pipes. As indicated, the rate of transfer of chemicals through each pipe is equal to a flow rate (Q, with units of cubic meters per second) multiplied by the concentration of the reactor from which the flow originates (c, with units of milligrams per cubic meter). If the system is at a steady state, the transfer into each reactor will balance the transfer out. Develop mass balance equations for the reactors and solve the three simultaneous
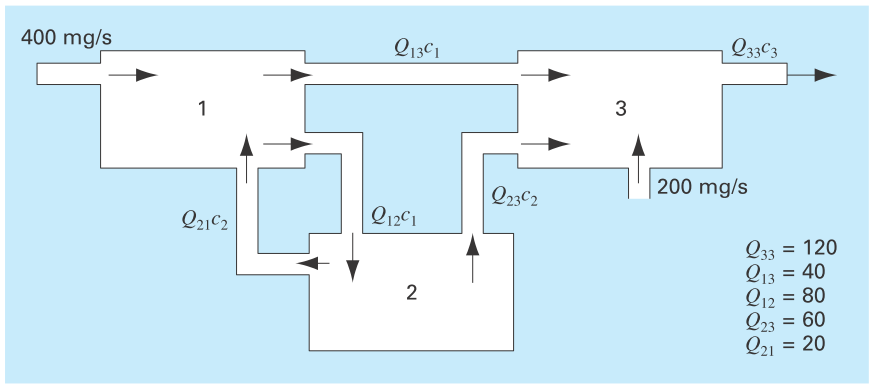
FIGURE P12.6
Three reactors linked by pipes. The rate of mass transfer through each pipe is equal to the product of flow Q and concentration c of the reactor from which the flow originates.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Numerical Methods for Engineers
Additional Math Textbook Solutions
Basic Technical Mathematics
Fundamentals of Differential Equations (9th Edition)
Advanced Engineering Mathematics
Finite Mathematics with Applications In the Management, Natural, and Social Sciences (12th Edition)
Calculus for Business, Economics, Life Sciences, and Social Sciences (14th Edition)
 Linear Algebra: A Modern IntroductionAlgebraISBN:9781285463247Author:David PoolePublisher:Cengage Learning
Linear Algebra: A Modern IntroductionAlgebraISBN:9781285463247Author:David PoolePublisher:Cengage Learning
