
Interpretation:
To determine the retrosynthetic analysis and synthesis of the following compounds.
Concept introduction:
Grignard reagents and organolithium compounds are strong bases. They will react with compounds containing hydrogen, attached to electronegative atoms like oxygen, nitrogen, and sulfur. They can be denoted as follows:
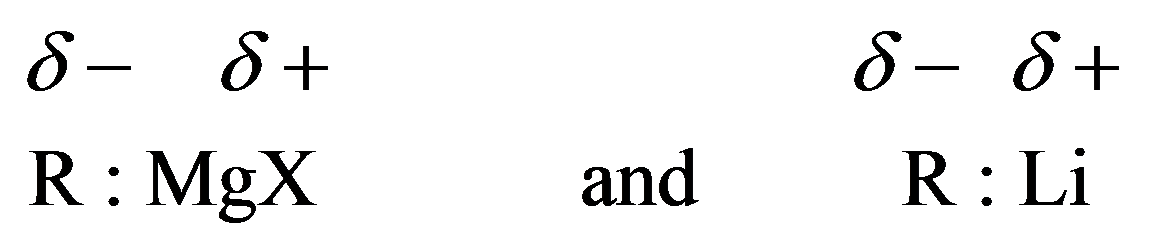
Grignard reagent reactions with carbonyl groups are treated as acid–base reactions. Grignard reagents are strong bases. When a reaction between a strong base and strong acid occurs, weak acid and weak base are formed. The side of the reaction that has the weak acid and base is favored.
Lithium aluminum hydride or sodium borohydride are used as reducing agents. Sodium borohydride is better for reducing
Retrosynthetic analysis is backtracking the reactants in a series of reactions. For synthesis of a compound using Grignard reagents, we have to select the correct reagents, ethers, aldehydes, ketones, or esters. There might be multiple ways to synthesize the alcohol and depending on the availability of reagents, we can choose a specific method.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
- Nucleophilic aromatic substitution provides one of the common methods for making phenols. ) Show how you would synthesize the following phenols, using benzene or toluene as your aromatic starting material, and explain why mixtures of products would be obtained in some cases. (a) m-cresol (b) p-n-butylphenolarrow_forward4. (A) A medicinal chemist wished to make a series of aromatic molecules bearing a ketone and thioether group with ortho, meta, or para relationships. To achieve this, they propose using nucleophilic aromatic substitution treating the corresponding ortho, meta, and para aryl chlorides with sodium ethanethiolate (NaSEt). Predict which of these reactions will likely work and which will likely fail. Provide a mechanistic explanation why. SET O NasEt EtS glagol Glal ol heat EtS target molecules EtS (B) Would the analogous reactions using EtMgBr instead of NaSEt be more or less likely to work? Explain why or why not. لمسلم EtMgBrarrow_forwardShow how the following compounds will be synthesized, beginning with benzene or toluene and any reagents required. Assume para, in ortho, para mixtures, are the main ingredients (and separable from ortho). 1) p-bromobenzene sulfonic acid 2) 1-phenypentane 3) m-chloronitrobenzene 4) o-chlorobenzoic acidarrow_forward
- Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short sequences of reactions that would be appropriate for each transformation. (a) 1,1,5-Trimethylcyclononane from 5,5-dimethylcyclononanonearrow_forwardStarting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.)p-Chlorobenzoic acidarrow_forwardStarting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.)m-Nitrobenzenesulfonic acidarrow_forward
- Nucleophilic aromatic substitution provides one of the common methods for making phenols. (Another method is discussedin Section 19-17.) Show how you would synthesize the following phenols, using benzene or toluene as your aromatic startingmaterial, and explain why mixtures of products would be obtained in some cases.(a) p-nitrophenol (b) 2,4,6-tribromophenolarrow_forwardGuiding your reasoning by retrosynthetic analysis, show how you could prepare each of the following compounds from the given starting material and any necessary organic or inorganic reagents. All require more than one synthetic step. (a) Cyclopentyl iodide from cyclopentane (b) 1-Bromo-2-methylpropane from 2-bromo-2-methylpropane (c) meso-2,3-Dibromobutane from 2-butyne (d) 1-Heptene from 1-bromopentane (e) cis-2-Hexene from 1,2-dibromopentane (f) Butyl methyl ether (CH3CH2CH2CH2OCH3) from 1-butenearrow_forwardNucleophilic aromatic substitution provides one of the common methods for making phenols. (Another method is discussedin Section 19-17.) Show how you would synthesize the following phenols, using benzene or toluene as your aromatic startingmaterial, and explain why mixtures of products would be obtained in some cases.(a) p-nitrophenol (b) 2,4,6-tribromophenol (c) p-chlorophenol(d) m-cresol (e) p-n-butylphenolarrow_forward
- a) Suggest a synthesis of the compound shown below. You are required to first do a retrosynthesis leading to commercially available starting material, and then the corresponding synthesis. In your synthesis, you may use any inorganic reagents you think I will assist in your synthesis, but can only use the following organic reagents; saturated hydrocarbon chain with 5 or less carbons and one functional group, unsaturated hydrocarbon chain with 5 or less carbons and one functional group in addition to the alkene or alkyne, saturated hydrocarbon ring with 6 or less carbons and one functional group, and unsaturated hydrocarbon ring with 6 or less carbons and one functional group. заarrow_forwardProvide methods for preparing the following through a Grignard synthesis.a) 3-methylbutanoic acidb) 2, 4-dinitrophenylacetic acidarrow_forwardDevise a synthesis for your assigned compound using organic reagents no larger than two or three carbons, or benzene, as your only organic source of carbon. That is, any organic compounds larger than three carbons (excluding benzene) required for your synthesis must be made as part of the solution to your problem. You may use any inorganic reagents necessary without restriction. Group 21 Structure: IN CH2CH3arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

