
Concept explainers
(a)
Interpretation:
Whether the following pair of molecule is constitutional isomers or not should be identified:
Concept Introduction:
Constitutional isomers can be defined as the pair of molecules with the same molecular formula, but different structural formula. This phenomenon is known as isomerism.
(b)
Interpretation:
Whether the following pair of molecule is constitutional isomers or not should be identified:
Concept Introduction:
Constitutional isomers can be defined as the pair of molecules with the same molecular formula, but different structural formula. This phenomenon is known as isomerism.
(c)
Interpretation:
Whether the following pair of molecule is constitutional isomers or not should be identified:
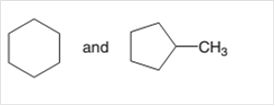
Concept Introduction:
Constitutional isomers can be defined as the pair of molecules with the same molecular formula, but different structural formula. This phenomenon is known as isomerism.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, & Biological Chemistry
- Arrange these compounds in order of increasing boiling point (values in C are 42, 24, 78, and 118). (a) CH3CH2OH (b) CH3OCH3 (c) CH3CH2CH3 (d) CH3COOHarrow_forwardAre the compounds in each pair constitutional isomers or are they not constitutional isomers of each other?arrow_forwardDraw all six ketone constitutional isomers of the formula C6H12O Follow the instructions and draw the five different possible arrangements of six carbon units. For parts A-C, draw only the six carbon atoms and Do Not draw the ketone. A:draw the structure containing six carbons in the longest parent chain. B: draw the two structures containing five carbons in the longest parent chain. C: Draw the two structures containing four carbons in the longest parent chain. D: Using the five structures from parts A,B,&C as a guide, draw the skeletal structures of the six constitutional isomers of C6H12O that are ketones.arrow_forward
- Classify the carbon atoms in each compound as 1°, 2°, 3°, or 4°. a. CH3CH2CH2CH3 b. (CH3)3CHarrow_forwardAre the compounds in each pair constitutional isomers or are they not isomers of each other?arrow_forwardTell whether the following pairs of compounds are identical, constitutional isomers, stereoisomers, or unrelated. (a) cis-1, 3-Dibromocyclohexane and trans-1, 4-dibromocyclohexane (b) 2, 3-Dimethylhexane and 2, 3, 3-trimethy1pentanearrow_forward
- 1. Draw two constitutional isomers that share the molecular formula C2H7P. Your structures will have the same molecular formula but will have different connectivities. 2. Draw two constitutional isomers that share the molecular formula C3H7Cl. Your structures will have the same molecular formula but will have different connectivities. 3. Draw two constitutional isomers that share the molecular formula C2H7N. Your structures will have the same molecular formula but will have different connectivities.arrow_forwardIs the following compound saturated or unsaturated: CH3CH2CH2CH2CH3 This compound is unsaturated because it has at least one double/triple bond between its carbons. This compound is unsaturated because it has all single bonds between its carbons. This compound is saturated because it only has one double/triple bond between its carbons. This compound is saturated because it has all single bonds between its carbons.arrow_forwardA2 1. An alkyne with molecular formula C5H10 2. A ketone with molecular formula C4H8O 3. A ketone with molecular formula C3H8O 4. An alkene with molecular formula C5H8 5. An alkene with molecular formula C5H10 6. An aldehyde with molecular formula C2H4O 7. An aldehyde with molecular formula CH4O 8. A saturated hydrocarbon with molecular formula C6H14arrow_forward
- Draw the structure of each molecule below and put an asterisk on each tetrahedral stereocenter. i) 3,4-dichlorohexane ii) 3-bromo-1-iodo-5-methylhexane iii) 3-bromo-2,4-dimethylpentanearrow_forwardDraw structures that t each description and name the functional group in each molecule: (a) two constitutional isomers with molecular formula C5H10O that contain different functional groups; (b) two constitutional isomers with molecular formula C6H10O that contain the same functional group.arrow_forward4. Which of the following has isomeric forms?a. C2H3Clb. C2H5Clc. C2HCld. C2H4Cl2 5. Which of the following hydrocarbons always gives the same product when one of its hydrogen atoms is replaced by a chlorine atom.a. Hexaneb. Hex-1-enec. Cyclohexaned. Cyclohexenearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


