
CHEMISTRY FDOC
13th Edition
ISBN: 9780134551272
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.4, Problem 12.36PP
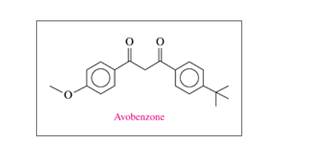
Avobenzone is a common ingredient in sunscreen. Its structural formula is shown.
a. What functional groups are in avobenzone?
b. What is the molecular formula and molar mass of avobenzone?
c. If a bottle of sunscreen containing 236 mL has 3.0% (m/v) avobenzone, how many grams of avobenzone are present?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
An extra strength Tylenol caplet contains 500 mg of acetaminophen (C8H9NO2) as one of its active ingredients.
a. How many Tylenol caplets will contain 0.050 mol of acetaminophen? _________ caplets
b. How many acetaminophen molecules do three caplets contain? __________________ molecules
1) How many mL of Propofol (10mg/ml) is needed to induce a 30kg dog for surgery at a dose of 6mg/kg?
2) How many mL of hydromorphine (2mg/ml) is necessary to use in a dog weighing 15kg at a dose of 0.2mg/kg?
3) How many mL of ivermectin (6mg/ml) should be given to a cat weighing 12.7 lbs at a dose of 0.024mg/kg?
4) How many mL of Midazolam (5mg/ml) should be given to a cat weighing 3.6 kg at a dose of 0.3mg/kg?
5) What dose should we give a puppy that weighs 4.5kg of Clavamox if we want to give it a dose of 6.25mg/lb?
a) 125mg
b) 62mg
c) 350mg
d) 165mg
6) How many mL of liquid Clindamycin (150mg/15ml) should be given to a 10lb kitten at a 16mg/kg dose?
7) Gatuno is a 7-year-old cat who came to the hospital because he has been vomiting for 3 days. He weighs 14.8lbs and is 8% dehydrated.
a) Calculate the lack of fluids.
b) We want to hydrate it for 5 hours.
8) Roxie is a puppy that weighs 8kg and is coming today for her teeth cleaning. Calculate Roxie's upkeep for her…
A sunscreen contains 6% oxybenzone and 3% avobenzone bymass. If a bottle contains 40. g of sunscreen, how many kilogramsof oxybenzone and avobenzone are needed to manufacture1 ton of sunscreen? How many bottles of sunscreen can beproduced for 1 ton of sunscreen?
Chapter 12 Solutions
CHEMISTRY FDOC
Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Prob. 12.3PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Give the common name for each of the following: a....Ch. 12.1 - Prob. 12.6PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Draw the condensed structural formula, or...Ch. 12.2 - Classify each of the following alcohols as primary...Ch. 12.2 - Classify each of the following alcohols as primary...
Ch. 12.2 - Prob. 12.11PPCh. 12.2 - Prob. 12.12PPCh. 12.2 - Give an explanation for each of the following...Ch. 12.2 - Give an explanation for each of the following...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Prob. 12.27PPCh. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Prob. 12.33PPCh. 12.4 - Prob. 12.34PPCh. 12.4 - Oxybenzone is an effective sunscreen whose...Ch. 12.4 - Avobenzone is a common ingredient in sunscreen....Ch. 12 - Prob. 12.37UTCCh. 12 - The compound frambinone has the taste of...Ch. 12 - A compound called resveratrol is an antioxidant,...Ch. 12 - A compound called cinnamaldehyde is found in...Ch. 12 - Prob. 12.41UTCCh. 12 - Prob. 12.42UTCCh. 12 - Prob. 12.43APPCh. 12 - Classify each of the following alcohols as primary...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Prob. 12.63CPCh. 12 - Draw the condensed structural formulas and give...Ch. 12 - A compound with the formula C4H8O is synthesized...Ch. 12 - A compound with the formula C5H10O oxidizes to...Ch. 12 - Compound A is a primary alcohol whose formula is...Ch. 12 - Compound X is a secondary alcohol whose formula is...Ch. 12 - Prob. 21CICh. 12 - Prob. 22CICh. 12 - Prob. 23CICh. 12 - Prob. 24CICh. 12 - Prob. 25CICh. 12 - lonone is a compound that gives violets their...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- C6H8O6(aq) + C12H7O2NCL2(aq) ----> C6H6O6(aq) + C12H9O2NCL2(aq) - C12H7O2NCL2 is abbreviated as DCIP - Molar Lass of C6H8O6(s) is 176.14 g/mol - Daily recommended intake (DRI) for adults: 90 mg of Vitamin C Askor & Bich found that it required 23.14 mlof 1.32 x 10^-3 M DCIP soliution to reach the endpoint when titrating a 7.50ml aliquot of pear juice. How many mg of ascorbic acid, C6H8O6 are present in the juice aliquot? _5.38_____(can you please show how to calculate this answer) How many ml of this juice would Bich have to drink in order to meet the daily recommended intake (DRI)?__126__ (arrow_forward3. Which is more effective as a disinfectant: ethyl alcohol or isopropyl alcohol? Choose only one and explain why. 4. What is the difference between cleaning, sanitizing, and disinfecting?arrow_forwardWhat volume of purified water, in milliliters, do you need to add to 5 mL promethazine 2 mg/mL solution to make promethazine 250 mcg/mL solution?arrow_forward
- what is 0.939g of 4-nitrobenzoyl chloride in mmol?arrow_forwardAnhydrous acetonitrile contains less than 0.001 wt% water. Expressthe water impurity in ppm.arrow_forwardScenario: you have a very bad head-ache so you run to the pharmacy to grab some medicine and are immediately over-whelmed with your options. You don’t have any allergies or sensitivities to any of the over the counter headache medicines, so you decide to buy the cheapest option to cure YOUR headache.Availability: Excedrin is available as a 200 caplet bottle for $15.51 and each caplet contains 250- mg of acetaminophen (the active ingredient in Excedrin). Advil is available as a 300 tablet bottle for $15.00 and each tablet contains 200-mg of ibuprofen (the active ingredient in Advil). Tylenol is available as a 225 pill bottle for $14.97 and each pill contains 500 mg of paracetamol (the active ingredient in Tylenol).Dosage Information: Paracetamol is dosed at 102 mg per 15 pounds of body mass. Ibuprofen is dosed at 113 mg per 25 pounds of body mass. Acetaminophen is dosed as 240 mg per 35 pounds of body mass.Active Ingredient Information: Acetaminophen has the chemical formula C8H9NO2.…arrow_forward
- How much cod liver oil is necessary to make 2500 capsules, each containing 0.33mL? a. 7.57 mL b. 825 mL c. 757 mL d. 0.0012mL e. 0.000012mLarrow_forwardtypical aspirin tablet contains 5.00 grains of pure aspirin analgesic compound. The rest of the tablet is starch. How many aspirin tablets can be made from 50.0 g of pure aspirin? [Use: 1.00 g = 15.4 grains] A) 17 tabletsarrow_forwardWhat would be the emperical formula for a compound that is made at STP from 3 mL H2 and 1 mL of N2.arrow_forward
- The identification of glucose in the drug (Glucose solution 20% -100 ml; hexamethylenetetramine 2.0 g) is carried out: A) Marku's reagent B) Tollens' reagent C) Dragendorff's reagent D) Fehling's reagentarrow_forward‘A’ compound works well with hard water. It is used for making shampoos & products for cleaning clothes. A is not 100% biodegradable and causes water pollution.‘B’ does not work well with hard water. It is 100% biodegradable and does not create water pollution. Identify A & B.arrow_forward1. Which is a better disinfectant, 70% or 95% ethyl alcohol? Why?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY