
Concept explainers
Lakes freeze from top down we all know that ice cubes float in a glass of water. Why? Virtually every substance contracts when it solidifies—the solid is denser than the liquid. If this happened to water. Ice cubes would sink to the bottom of a glass, and ice sheets would sink to the bottom of a lake. Fortunately, this doesn't happen. Liquid water expands by
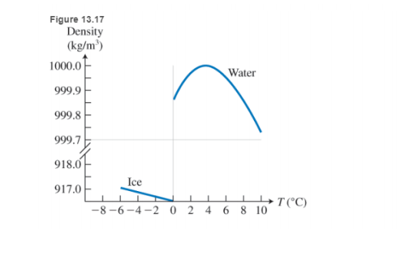
But this is not the only special thing about water. While the density of most substances increases when they are cooled, water density shows a very peculiar temperature dependence (see Figure 13.17). As the temperature decreases, water density increases, but only until
Note that if water were most dense at the freezing point, then in the winter the very cold water at the surface of lakes would sink in this case the lake would freeze from the bottom up, and an life in it would be destroyed.
The expansion of water when it freezes has another important environmental benefit the so-called freeze-thaw effect on sedimentary rocks. Water is absorbed into cracks in these rocks and men freezes in cold weather. The solid ice expands and cracks me rock like a wood-cutter splitting logs. This continual process of liquid water absorption, freezing, and cracking releases mineral and nitrogen deposits into me soil and can eventual break the rock down into soil.
Using Newton’s second law, expressions for buoyant force and other forces, and the densities of liquid and solid water at
a. 0.84
b. 0.88
c. 0.92
d. 0.96
e. 1.00
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
College Physics
Additional Science Textbook Solutions
Essential University Physics: Volume 2 (3rd Edition)
College Physics
Lecture- Tutorials for Introductory Astronomy
Applied Physics (11th Edition)
Physics: Principles with Applications
College Physics: A Strategic Approach (3rd Edition)
- An ice cube is placed in a glass of water. What happens to the level of the water as the ice melts?arrow_forwardBird bones have air pockets in them to reduce their weight—this also gives them an average density significantly less than that of the bones of other animals. Suppose an ornithologist weighs a bird bone in air and in water and finds its mass is 45.0 g and its apparent mass when submerged is 3.60 g (the bone is watertight). (a) What mass of water is displaced? (b) What is the volume of the bone? (c) What is its average density?arrow_forward. Air flows through a heating duct with a square cross-section with 8-inch sides at a speed of 5.0 ft/s. Just before reaching an outlet in the floor of a room, the duct widens to assume a square cross-section with sides equal to 12 inches. Compute the speed of the air flowing into the room, assuming that we can treat the air as an incompressible fluid.arrow_forward
- You observe two helium balloons floating next to each other at the ends of strings secured to a table. The facing surfaces of the balloons are separated by 1-2 cm. You blow through the small space between the balloons. What happens to the balloons? (a) They move toward each other. (b) They move away from each other. (c) They are unaffected.arrow_forwardMany figures in the text show streamlines. Explain why fluid velocity is greatest where streamlines are closest together. (Hint: Consider the relationship between fluid velocity and the cross-sectional area through which it flows.)arrow_forwardAt a fraternity party, drinking straws have been joined together to make a giant straw that will be used to drink punch placed in a bowl on the ground from atop the fraternity house building. What is the maximum allowable height of the building if the partygoers are successful in drinking the punch? Assume the density of the punch is the same as the density of water.arrow_forward
- Explain why the viscosity of a liquid decreases with temperature, that is, how might an increase in temperature reduce the effects of cohesive forces in a liquid? Also explain why the viscosity of a gas increases with temperature, that is, how does increased gas temperature create more collisions between atoms and molecules?arrow_forward. A dentist's chair with a person in it weighs 1900 N. The output plunger of a hydraulic system starts Do lift the chair when the dental assistant's foot exerts a force of 45 N on the input piston. Neglecting any difference in the heights of the piston and the plunger, what is the ratio of the area of the plunger to the area of the piston?arrow_forwardToe dancing (as in ballet) is much harder on toes than normal dancing or walking. Explain in terms of pressure.arrow_forward
- Some miners wish to remove water from a mine shaft. A pipe is lowered to the water 90 m below, and a negative pressure is applied to raise the water. (a) Calculate the pressure needed to raise the water. (b) What is unreasonable about this pressure? (c) What is unreasonable about the premise?arrow_forwardHeroes in movies hide beneath water and breathe through a hollow reed (villains never catch on to this trick). In practice, you cannot inhale in this manner if your lungs are more than 60.0 cm below the surface. What is the maximum negative gauge pressure you can create in your lungs on dry land, assuming you can achieve -3.00 cm water pressure with your lungs 60.0 cm below the surface?arrow_forwardAn apple is held completely submerged just below the surface of a container of water. The apple is then moved to a deeper point in the water. Compared with the force needed to hold the apple just below the surface, what is the force needed to hold it at a deeper point? (a) larger (b) the same (c) smaller (d) impossible to determinearrow_forward
 College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University
University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





