
(a)
Interpretation:
It is to be shown how a retrosynthetic analysis might be constructed for the given synthesis.
Concept introduction:
Retrosynthesis is the planning of
Answer to Problem 13.30P
The retrosynthesis for the given synthesis is
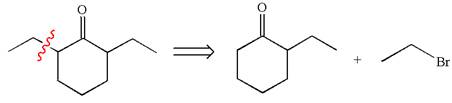
Explanation of Solution
The given synthetic reaction is
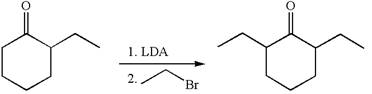
In the given synthesis, the product differs from the starting compound by one ethyl group bonded to six-membered ring. Thus, the bond between the ethyl group and ring carbon in the target must break to transform it to the starting compound.
Therefore, retrosynthesis for the given synthetic reaction is
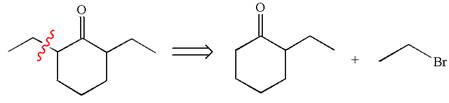
The retrosynthesis for the given synthetic reaction is shown by disconnecting the ethyl group from the ring.
(b)
Interpretation:
It is to be shown how a retrosynthetic analysis might be constructed for the given synthesis.
Concept introduction:
Retrosynthesis is the planning of organic synthesis, working backwards from target molecule to a simpler precursor, regardless of any interaction with reagents. Thus, the basis of retrosynthetic analysis is the transform, which means the reverse of a synthetic reaction. The precursors are the compounds, which are either readily available or easy to produce. The transform is indicated by an open arrow
Answer to Problem 13.30P
The retrosynthesis for the given synthesis is
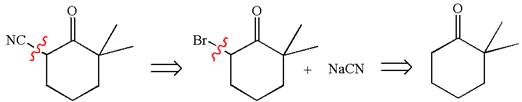
Explanation of Solution
The given synthetic reaction is
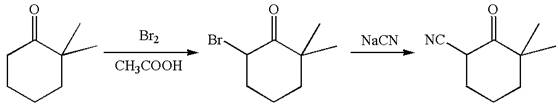
The given synthesis is of two steps; the final product is the target molecule. Thus, retrosynthesis could be planned from the target to the intermediate product to the starting material. The target molecule and the intermediate product differ in bromine and cyanide. Thus, the bond between the cyanide and ring carbon must break to transform into an intermediate. The intermediate and the starting molecule differ by bromine atom. Thus, the bond between the bromine and ring carbon must break to transform into the starting material.
Therefore, the retrosynthesis for the given synthetic reaction is
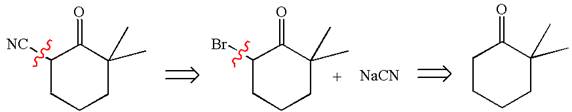
The retrosynthesis for the given synthetic reaction is shown by disconnecting the cyanide group and then by disconnecting the bromine atom from the ring.
(c)
Interpretation:
It is to be shown how a retrosynthetic analysis might be constructed for the given synthesis.
Concept introduction:
Retrosynthesis is the planning of organic synthesis, working backwards from target molecule to a simpler precursor, regardless of any interaction with reagents. Thus, the basis of retrosynthetic analysis is the transform, which means the reverse of a synthetic reaction. The precursors are the compounds, which are either readily available or easy to produce. The transform is indicated by an open arrow
Answer to Problem 13.30P
The retrosynthesis for the given synthesis is
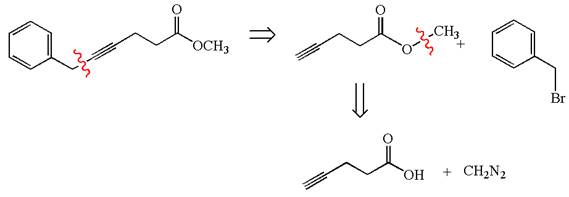
Explanation of Solution
The given synthetic reaction is
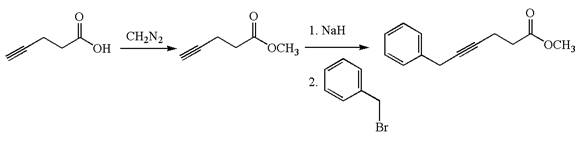
The given synthesis is of two steps; the final product is the target molecule. Thu, s the retrosynthesis could be planned from the target to the intermediate product to the starting material. The target molecule and the intermediate product differ in the benzyl group attached to the triple bonded carbon. Thus, the bond between the benzylic carbon and triple bonded carbon must break to transform into an intermediate. The intermediate can be transformed into the starting material by replacing the methyl group bonded to the oxygen atom by hydrogen.
Therefore, the retrosynthesis for the given synthetic reaction is
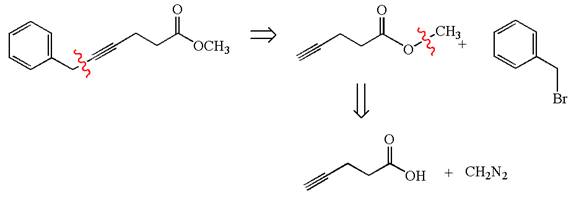
The retrosynthesis for the given synthetic reaction is shown by disconnecting the benzyl group from the triple bonded carbon and then by disconnecting the methyl group from the oxygen atom.
(d)
Interpretation:
It is to be shown how a retrosynthetic analysis might be constructed for the given synthesis.
Concept introduction:
Retrosynthesis is the planning of organic synthesis, working backwards from target molecule to a simpler precursor, regardless of any interaction with reagents. Thus, the basis of retrosynthetic analysis is the transform, which means the reverse of a synthetic reaction. The precursors are the compounds, which are either readily available or easy to produce. The transform is indicated by an open arrow
Answer to Problem 13.30P
The retrosynthesis for the given synthesis is
Explanation of Solution
The given synthetic reaction is
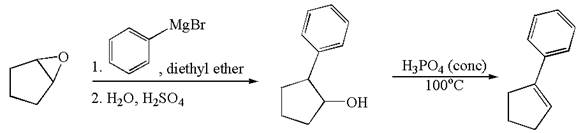
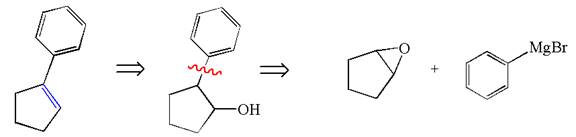
The given synthesis is of two steps; the final product is the target molecule. Thus the retrosynthesis could be planned from the target to the intermediate product to the starting material. The target molecule has a double bond, which is removed in the intermediate product having the ydroxyl group at that position. Thus, the
Therefore, the retrosynthesis for the given synthetic reaction is
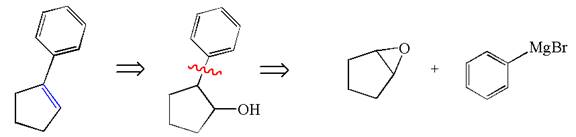
The retrosynthesis for the given synthetic reaction is shown by disconnecting the
(e)
Interpretation:
It is to be shown how a retrosynthetic analysis might be constructed for the given synthesis.
Concept introduction:
Retrosynthesis is the planning of organic synthesis, working backwards from target molecule to a simpler precursor, regardless of any interaction with reagents. Thus, the basis of retrosynthetic analysis is the transform, which means the reverse of a synthetic reaction. The precursors are the compounds, which are either readily available or easy to produce. The transform is indicated by an open arrow
Answer to Problem 13.30P
The retrosynthesis for the given synthesis is
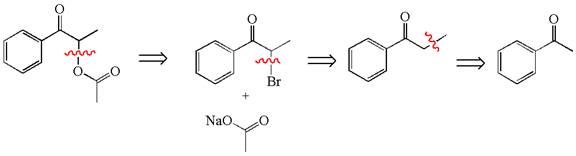
Explanation of Solution
The given synthetic reaction is
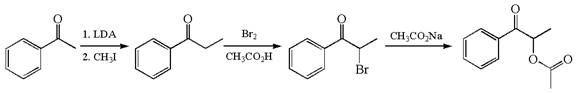
The given synthesis is of three steps; the final product is the target molecule. Thus, the retrosynthesis could be planned from the target to the second intermediate, then to the first intermediate, and finally into the starting material. The target molecule has the acetate group at the alpha position and the second intermediate has the bromine atom; thus, the bond between the alpha carbon and the oxygen of the acetate group must break. The second intermediate can be transformed into the first intermediate by breaking the bond between bromine and the alpha carbon. The first intermediate and the starting molecule differ by an additional methyl group; thus the bond between the alpha carbon and methyl must break to show the transform.
Therefore, the retrosynthesis for the given synthetic reaction is
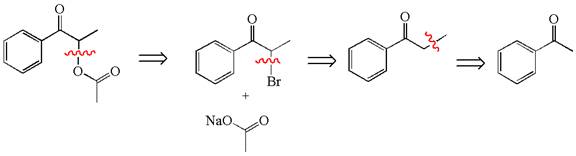
The retrosynthesis for the given synthetic reaction is shown by disconnecting appropriate bonds.
(g)
Interpretation:
It is to be shown how a retrosynthetic analysis might be constructed for the given synthesis.
Concept introduction:
Retrosynthesis is the planning of organic synthesis, working backwards from target molecule to a simpler precursor, regardless of any interaction with reagents. Thus, the basis of retrosynthetic analysis is the transform, which means the reverse of a synthetic reaction. The precursors are the compounds, which are either readily available or easy to produce. The transform is indicated by an open arrow
Answer to Problem 13.30P
The retrosynthesis for the given synthesis is
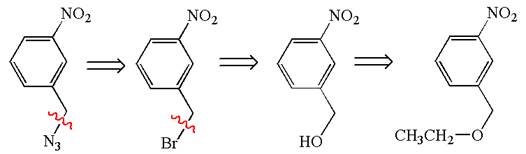
Explanation of Solution
The given synthetic reaction is
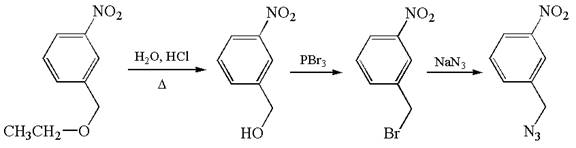
The given synthesis is of three steps; the final product is the target molecule. Thus; the retrosynthesis could be planned from the target to the second intermediate; then to the first intermediate; and finally into the starting material. The target can be transformed to the second intermediate by replacing the nitride group by bromine; thus the bond between the nitride group and carbon must break. The second intermediate can be transformed into the first intermediate by replacing the bromine by hydroxyl group; thus the bond between bromine and carbon must break. The first intermediate can be transformed into the starting material by replacing the hydroxyl group by ethoxy group.
Therefore, the retrosynthesis for the given synthetic reaction is
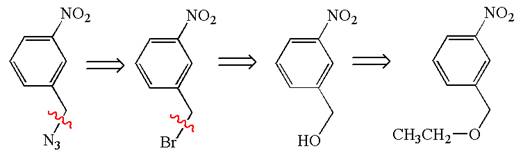
The retrosynthesis for the given synthetic reaction is shown by disconnecting appropriate bonds.
Want to see more full solutions like this?
Chapter 13 Solutions
Get Ready for Organic Chemistry
- 15) Provide a detailed, stepwise mechanism for the acid-catalyzed condensation reaction between benzaldehyde and methylamine.arrow_forwardIf possible would you be able to solve and explain these questions?arrow_forwardThe two reactions that follow involve nucleophilic substitution at an acyl carbon. For each reaction, show the electrophile and the nucleophile in the key bond- forming step, the corresponding tetrahedral addition intermediate, and the leaving group in the bond-breaking step. Write a general mechanistic scheme (use curved arrows to show bond-making and bond-breaking processes) for these reactions, keeping in mind that acid- and base-catalyzed processes will differ in their timing of proton transfers. 00 || || PhCOCPh+ C₂H5OH 00 |||| PhCOCPh+ 2 CH3NH H₂SO4 cat. O PhCOC₂H5+ PhCO₂H PhCNCHg + CH3NH PhCOarrow_forward
- Please don't provide handwritten solution ....arrow_forwardProvide a retrosynthetic analysis and forward synthesis for the compound shown below. H- and any other reagent of one carbon or less.arrow_forwardThe following two isomeric ketones were among the 68 compounds isolated from the steam-distilled volatile oil of fresh and air-dried marijuana buds (). Nat. Prod. 1996, 59, 49-51). Propose a separate synthesis for each of these two compounds using only disubstituted alkenes containing four carbon atoms as starting materials. 12.75a Of the following, which represents a logical retrosynthesis for target molecule A? MgBr OH MgBr MgBr OH OH MgBrarrow_forward
- a) Suggest a synthesis of the compound shown below. You are required to first do a retrosynthesis leading to commercially available starting material, and then the corresponding synthesis. In your synthesis, you may use any inorganic reagents you think I will assist in your synthesis, but can only use the following organic reagents; saturated hydrocarbon chain with 5 or less carbons and one functional group, unsaturated hydrocarbon chain with 5 or less carbons and one functional group in addition to the alkene or alkyne, saturated hydrocarbon ring with 6 or less carbons and one functional group, and unsaturated hydrocarbon ring with 6 or less carbons and one functional group. заarrow_forwardHow does the reactivity of an aldehyde or a ketone toward nucleophiles compare with thereactivity of the carbonyl compounds whose reactions you studied in Chapter 15?arrow_forwardThe reaction that occurs when the benzaldehyde you have is reacted in a basic environment is called the Cannizzaro reaction, and when it is reacted with cyanide, it is called benzoin. It Explain the reason by writing the reaction mechanisms of the reactions.arrow_forward
- 1.) Design a detailed synthetic route to prepare the following compound using cyclopentane, cyclohexanone, cyclohexan-1,3-dione, benzene, phenol, diethyl malonate, ethyl acetoacetate, as the starting materials, and any other reagents of less than 5 carbon atoms (including 5 carbon atoms). O OEtarrow_forwardAldol Condensation- Synthesis of Dibenzalacetone The protocol says that, after adding in all the reactants, stir for an additional 15 minutes. A student swirled for only 8 minutes and then, correctly, stopped and proceeded with isolating the product. What did the student do that gave such confidence and accuracy?arrow_forwardProvide a stepwise synthesis for benzyl alcohol to sodium benzoate with reagents KMnO4 and NaOH (including arrow-pushing mechanisms) Please show all the arrow push with the given reagents above.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

