
Concept explainers
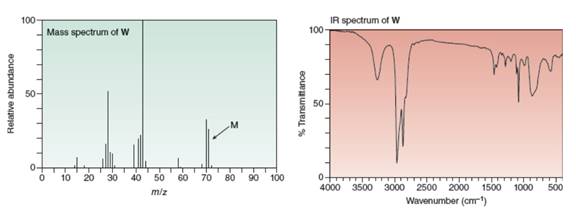
Reaction of
gives the IR and mass spectra shown below. Propose a structure for
a stepwise mechanism that accounts for its formation.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
ALEKS 360 CHEMISTRY ACCESS
Additional Science Textbook Solutions
Chemistry: The Molecular Nature of Matter
Chemistry (7th Edition)
Introduction to Chemistry
Inorganic Chemistry
Fundamentals of Heat and Mass Transfer
Chemistry: The Central Science (13th Edition)
- an unknown compound of molecular formula C4H10O only showed singlets in its 1H-NMRarrow_forwardTreatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forwardTwo products are obtained from the reaction of (Z)-pent-2-ene with water in sulfuric acid. The mass spectra of these products are shown below. Draw the structure of each product, and match the product to its mass spectrumarrow_forward
- Choose the structure corresponding to the given 1H and 13C NMR spectraarrow_forwardCompound A, MW 86, shows an IR absorption at 1730 cm-1 and a very simple 1H NMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.arrow_forwardTreatment of 3,4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how you would use 1H and 13CNMR spectroscopy to help identify them. How would you use UV spectroscopy?arrow_forward
- One way to determine the number of acidic hydrogens in a molecule is to treat the compound with NaOD in D2O, isolate the product, and determine its molecular weight by mass spectrometry. For example, if cyclohexanone is treated with NaOD in D2O, the product has MW = 102. Explain how this method works.arrow_forwardAnethole, C10H12O, a major constituent of the oil of anise, has the 1H NMR spectrum shown. On oxidation with Na2Cr2O7, anethole yields p-methoxybenzoic acid. What is the structure of anethole? Assign all peaks in the NMR spectrum, and account for the observed splitting patterns.arrow_forward3-Chlorocyclopropene, on treatment with AgBF4, gives a precipitate of AgCl and a stable solution of a product that shows a single 1H NMR absorption at 11.04 δ. What is a likely structure for the products, and what is its relation to HĂ¼ckel’s rule?arrow_forward
