
ORG CHEM CONNECT CARD
6th Edition
ISBN: 9781264860746
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 38P
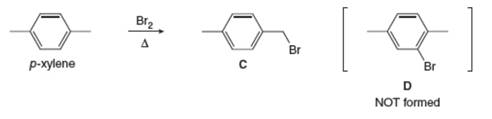
Explain why radical bromination of p-xylene forms
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Can you explain the process of free radical bromination by using methylcyclohexane?
The Friedel-Crafts Alkylation of p-xylene n-propyl bromide can result in an isopropyl as well as n-propyl substitution.
A) Write the chemical equation involved in this reaction.
B) Which will be the major and minor product? What do you expect will be the ratio of n-propyl to isopropyl substitution for p-xylene?
Explain why the addition of HBr to alkenes A and C is regioselective, forming addition products B and D, respectively.
Chapter 13 Solutions
ORG CHEM CONNECT CARD
Ch. 13.1 - Prob. 1PCh. 13.1 - Prob. 2PCh. 13.2 - Prob. 3PCh. 13.3 - Prob. 4PCh. 13.3 - Prob. 5PCh. 13.4 - Prob. 7PCh. 13.5 - Problem 15.8 Which bond in the each compound is...Ch. 13.6 - Prob. 9PCh. 13.6 - Prob. 10PCh. 13.7 - Prob. 11P
Ch. 13.7 - Prob. 12PCh. 13.8 - Prob. 13PCh. 13.8 - Prob. 14PCh. 13 - Prob. 27PCh. 13 - Prob. 28PCh. 13 - Prob. 34PCh. 13 - 15.37 What alkane is needed to make each alkyl...Ch. 13 - 15.38 Which alkyl halides can be prepared in good...Ch. 13 - Prob. 37PCh. 13 - 15.40 Explain why radical bromination of p-xylene...Ch. 13 - a. What product(s) (excluding stereoisomers) are...Ch. 13 - Prob. 40PCh. 13 - 15.43 Draw the products formed when each alkene is...Ch. 13 - 15.44 Draw all constitutional isomers formed when...Ch. 13 - 15.45 Draw the organic products formed in each...Ch. 13 - Prob. 45PCh. 13 - 15.47 Treatment of a hydrocarbon A (molecular...Ch. 13 - 15.48 Draw the products formed in each reaction...Ch. 13 - 15.53 Consider the following bromination: .
a....Ch. 13 - 15.54 Draw a stepwise mechanism for the following...Ch. 13 - Prob. 57PCh. 13 - 15.57 Devise a synthesis of each compound from...Ch. 13 - Prob. 59PCh. 13 - Prob. 60PCh. 13 - 15.60 Devise a synthesis of each compound using ...Ch. 13 - Prob. 62PCh. 13 - Prob. 63PCh. 13 - 15.63 As described in Section 9.16, the...Ch. 13 - 15.64 Ethers are oxidized with to form...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structure of the minor product formed in the reaction of 2-methylbut-1-ene with concentrated sulfuric acid Explain why thid is the major product? Structure of minor product? Explanation?arrow_forwardExplain why the addition of HBr to alkenes A and C is regioselective,forming addition products B and D, respectively.arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 CH3 CHCCH3 OH CH3 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. Sn [F ChemDoodlearrow_forward
- Draw the alkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 CH3CCH3 OH You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. Sn [F ChemDoodleⓇarrow_forwardRank the following alkenes from least to most stable. Then explain why you picked this order.arrow_forwardExplain the mechanism for halogenation of alkenes ?arrow_forward
- Draw all monochlorination products obtained from the free radical chlorination of 2,3,5-trimethylhexane (include all stereoisomers)arrow_forward1. What are the various ways by which alkenes may be synthesized?2. Give two examples each of Unsymmetrical alkenes and reagents.3. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition productsarrow_forwardWhy does 2,3-dimethyl-2-butene react faster than 2-methyl-2-butene?arrow_forward
- Identify the substitution products that form when 2-bromo-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water. a. Explain why the same products are obtained when 2-chloro-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.arrow_forwardRadical bromination of propene using NBS gives 3-bromo-1-propene. Draw the allylic radical intermediate formed during this reaction, showing both resonance structures.arrow_forwardExplain Halogenation of Activated Benzenes ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License