
Concept explainers
Interpretation:
According to the terpene rule, the structure of caryophyllene needs to be explained.
Concept introduction:
Terpene rule or isoprene rule states that terpenoids structures are obtained by linking isoprene unit. Isoprene (2-methyl-1, 3-butadiene) is the monomer of natural rubber.
Answer to Problem 3Q
The structure of caryophyllene fits in the terpene rule as caryophyllene contains a moiety which is similar to the polyisoprene unit.
Explanation of Solution
The structure of caryophyllene is represented as follows:

The
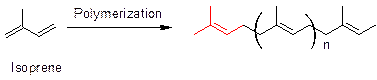
Polymerization of isoprene leads to a basic structure that is present in all terpenoids (shown in red). Similarly, caryophyllene also has a similar moiety (shown red) as present in terpenoids. Thus, the structure of caryophyllene fits in the terpene rule.
Want to see more full solutions like this?
Chapter 13 Solutions
SMALL SCALE APP.TO ORG...(LL) >CUSTOM<
- 2) During the washing of 2 - methyl - 2- butanol and HCL with sodium bicarbonate, a reaction can occur which leads to te formation of a lower boiling by peoduct. Considering the nature of sodium bicarbonate, suggest a possible reaction and structure for the product.arrow_forwardWrite the mechanism and show the product of the reaction of 3- pentanone with n-butylamine with the molecule below with a catalytic amount of acid:arrow_forwardIn addition to the normal dangers in using concentrated hydrochloric acid, what additional dangers are presented in mixing it with ice and what precautions should be taken? What gas is evolved when hydrochloric acid is added to the borohydride reduction with benzophenone?arrow_forward
- Although derivatization of a reaction should be avoided as much as possible according to the principles of green chemistry, the protective groups needed in organic synthesis mainly regulate the chemoselectivity of the reaction. Mention only 3 requirements for a good protective group !arrow_forwardSuggest a simple product that is related to Organic Chemistry that easily can do at home. Show some reactions and mechanisms.arrow_forwardRegarding an organic chemistry laboratory experiment: a. Why is it necessary to isolate eugenol from cloves using (direct) steam distillation rather simple distillation? b. In this experiment, why does eugenol distill at a temperature far below its boiling point? c. Why is diethyl ether used to extract eugenol from the distillate rather than ethanol?arrow_forward
- Sodium hydroxide was used to help separate benzoic acid and fluorenone because deprotonated benzoic acid (sodium benzoate) favorably partitions into the aqueous phase (and fluorenone does not). Would this procedure have worked without the sodium hydroxide (using simply neutral water)? Why or why not?arrow_forwardWhat would be the drawing for these mechanisms?arrow_forwardQ1: Give one lysis buffer that is commonly used for western blotting experiments and include its components Q3: To make sure that you used a similar amount of samples, what important step should be done before proceeding the electrophoresis stage? Q4. Why is it necessary to store the prepared lysates in a very low temperature?arrow_forward
- 1. You have been given a mixture of toluene,aniline and benzoic acid dissolved in chloroform.explain in detail with the aid of a flow diagram and reaction schemes,how you would separate the mixture into pure componentsarrow_forwardWhy must the stopper at the top of the separatory funnel be removed before liquid can be withdrawn through the stopcock? - Write four equations (showing structures) for the reactions used in this experiment to separate and retrieve \ ( \mathrm {p} \)-bromoaniline and benzoic acid.arrow_forwardThe experiment involves Friedel- Crafts acylation. How can sulfuric acid be used as an alternative instead of zinc chloride as a catalyst? Explainarrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

