
ORGANIC CHEMISTRY W/CONNECT & ALEKS
6th Edition
ISBN: 9781264683888
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 64P
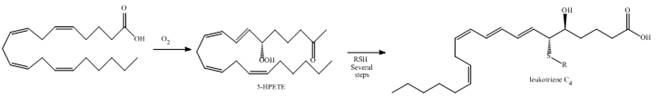
As described in Section 9.16, the leukotrienes, important components in the asthmatic response, are synthesized from arachidonic acid via the hydroperoxide
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Use the priority system to draw the structure of the following molecule:
2-methoxy-3-methyl-5-oxoheptanoic acid
Aspirin is an anti-inammatory agent because it inhibits the conversion of arachidonic acid to prostaglandins by the transfer of its acetyl group (CH3CO –) to an OH group at the active site of an enzyme (Section 19.6). This reaction, called transesterication, results in the conversion of one ester to another by a nucleophilic acyl substitution reaction. Draw a stepwise mechanism for the given transesterication.
Prostaglandins are a class of eicosanoids, fatty acid derivatives with a variety of extremely potent actions on vertebrate tissues.
They are responsible for producing fever and inflammation and its associated pain. Prostaglandins are derived from the 20-
carbon fatty acid arachidonic acid in a reaction catalyzed by the enzyme prostaglandin endoperoxide synthase. This enzyme, a
cyclooxygenase, uses oxygen to convert arachidonic acid to PGG2, the immediate precursor of many different prostaglandins.
Rate of formation of PGG2 with 10 mg/ml
ibuprofen (mM/min)
Arachidonic
acid (mM)
Rate of formation
of PGG2 (mM/min)
0.190
12.3
0.228
13.9
0.342
17.5
0.570
1.33
22.2
28.8
7.71
8.88
11.9
16.3
24.0
The kinetic data given in the table are for the reaction catalyzed by a mutant of prostaglandin endoperoxide synthase. Focusing
here on the first two columns, determine the Vmax and Km of the enzyme.
Vmax
Km
=
mM/min
mM
Chapter 13 Solutions
ORGANIC CHEMISTRY W/CONNECT & ALEKS
Ch. 13.1 - Prob. 1PCh. 13.1 - Prob. 2PCh. 13.2 - Prob. 3PCh. 13.3 - Prob. 4PCh. 13.3 - Prob. 5PCh. 13.4 - Prob. 7PCh. 13.5 - Problem 15.8 Which bond in the each compound is...Ch. 13.6 - Prob. 9PCh. 13.6 - Prob. 10PCh. 13.7 - Prob. 11P
Ch. 13.7 - Prob. 12PCh. 13.8 - Prob. 13PCh. 13.8 - Prob. 14PCh. 13 - Prob. 27PCh. 13 - Prob. 28PCh. 13 - Prob. 34PCh. 13 - 15.37 What alkane is needed to make each alkyl...Ch. 13 - 15.38 Which alkyl halides can be prepared in good...Ch. 13 - Prob. 37PCh. 13 - 15.40 Explain why radical bromination of p-xylene...Ch. 13 - a. What product(s) (excluding stereoisomers) are...Ch. 13 - Prob. 40PCh. 13 - 15.43 Draw the products formed when each alkene is...Ch. 13 - 15.44 Draw all constitutional isomers formed when...Ch. 13 - 15.45 Draw the organic products formed in each...Ch. 13 - Prob. 45PCh. 13 - 15.47 Treatment of a hydrocarbon A (molecular...Ch. 13 - 15.48 Draw the products formed in each reaction...Ch. 13 - 15.53 Consider the following bromination: .
a....Ch. 13 - 15.54 Draw a stepwise mechanism for the following...Ch. 13 - Prob. 57PCh. 13 - 15.57 Devise a synthesis of each compound from...Ch. 13 - Prob. 59PCh. 13 - Prob. 60PCh. 13 - 15.60 Devise a synthesis of each compound using ...Ch. 13 - Prob. 62PCh. 13 - Prob. 63PCh. 13 - 15.63 As described in Section 9.16, the...Ch. 13 - 15.64 Ethers are oxidized with to form...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 17-29 Why can’t two molecules of acetone form a hydrogen bond with each other?arrow_forwardWhich carboxylic acid has salts that are used to inhibit yeast and mold growth in the following? a. Ketchup and syrup b. Sauerkraut c. Pie filling d. Baked productsarrow_forwardDraw structural formula of the products formed from the following reactions: reduction of acetone acetophenone + H2 , Ni 3-hydroxypentanal + Tollen's reagentarrow_forward
- SYNTHESIS OF ESTERS VIA NUCLEOPHILIC ACYL SUBSTITUTION Write the chemical equation involved in the reaction between the excess acid and NaHCO3. Explain why NaHCO3 is preferred over NaOH for the neutralization of excess acid. How was excess alcohol eliminated from the crude product.arrow_forwardDescribe the reaction where benzil is prepared by the oxidation of an a-hydroxyketone, benzoin and uses nitric acid to preform oxidation.arrow_forwardChoose among these options Among these Inhibits the enzyme which is responsible for the assembly of matured viral particles Saquinavir Didanosine Penciclovir Azidothymidine The following are essential components in the structure of sulfonamides with the exception of Amino group Benzene ring Pyridine N-Acyl amino This drug has been associated with optic neuritis and therefore discontinued after 2 months of treatment Isonizaid Rifampicin Ethambutol Pyrazinamide A non prodrug ACE Inhibitor Captopril Enalapril Quinapril Fosinopril High sodium diet High fat diet Low potassium diet Low phosphate dietarrow_forward
- What is the role of concentrated H2SO4 in the esterification reaction?arrow_forwardwrite an equation showing the formation of a y-lactone from a 5-carbon hydroxy carboxylic acidarrow_forwardWhat reagent serves as the source of H₂S? Oxalic Acid Ammonium Hydroxide Sulfur Acetate Thioacetamidearrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY