
ORGANIC CHEMISTRY-WILEYPLUS NEXTGEN
4th Edition
ISBN: 9781119760924
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 13, Problem 73IP
Interpretation Introduction
Interpretation: The mechanism for the given transformation needs to be provided and stereochemistry needs to be explained.
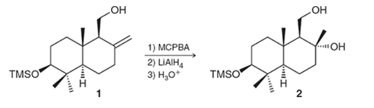
Concept Introduction:
In the presence of MCPBA, the formation of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Show how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any necessary reagents.m-toluonitrile(a) CH3 C N CH3 CH2NH2m-methylbenzylamine(b)m-iodotoluene
Show how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any necessary reagents.m-toluonitrile(a) CH3 C N CH3 CH2NH2m-methylbenzylamine(b)m-iodotolueneCH I 3 (c)m-cresol(d) CH3 OH3-methyl-4-nitroanilineCH3 NH2O2N(e)NHCH3N-cyclopentyl-m-toluidine
Identify the reagent that accomplish the transformation seen below:
1) 03
2) Zn/H₂O
H₂O/H2SO4 (cat.)
OH₂/Pd
O
1) BH3
2) H₂O2/NaOH / H₂O
m-CPBA (meta-chloroperoxybenzoic acid)
HO
Chapter 13 Solutions
ORGANIC CHEMISTRY-WILEYPLUS NEXTGEN
Ch. 13.2 - Prob. 1LTSCh. 13.2 - Prob. 1PTSCh. 13.2 - Prob. 2PTSCh. 13.2 - Prob. 3ATSCh. 13.4 - Prob. 4CCCh. 13.5 - Prob. 2LTSCh. 13.5 - Prob. 5PTSCh. 13.5 - Prob. 6ATSCh. 13.5 - Prob. 7CCCh. 13.5 - Prob. 8CC
Ch. 13.5 - Prob. 9CCCh. 13.6 - Prob. 10CCCh. 13.7 - Prob. 11CCCh. 13.7 - Prob. 12CCCh. 13.8 - Prob. 3LTSCh. 13.8 - Prob. 13PTSCh. 13.8 - Prob. 14ATSCh. 13.9 - Prob. 15CCCh. 13.10 - Prob. 4LTSCh. 13.10 - Prob. 17ATSCh. 13.10 - Prob. 5LTSCh. 13.10 - Prob. 19ATSCh. 13.11 - Prob. 20CCCh. 13.12 - Prob. 6LTSCh. 13.12 - Prob. 7LTSCh. 13 - Prob. 26PPCh. 13 - Prob. 27PPCh. 13 - Prob. 28PPCh. 13 - Prob. 29PPCh. 13 - Prob. 30PPCh. 13 - Prob. 31PPCh. 13 - Prob. 32PPCh. 13 - Prob. 33PPCh. 13 - Prob. 34PPCh. 13 - Prob. 35PPCh. 13 - Prob. 36PPCh. 13 - Prob. 37PPCh. 13 - Prob. 38PPCh. 13 - Prob. 39PPCh. 13 - Prob. 40PPCh. 13 - Prob. 41PPCh. 13 - Prob. 42PPCh. 13 - Prob. 43PPCh. 13 - Prob. 44PPCh. 13 - Prob. 45PPCh. 13 - Prob. 46ASPCh. 13 - Prob. 47ASPCh. 13 - Prob. 48ASPCh. 13 - Prob. 49ASPCh. 13 - Prob. 50ASPCh. 13 - Prob. 51ASPCh. 13 - Prob. 52ASPCh. 13 - Prob. 53ASPCh. 13 - Prob. 54IPCh. 13 - Prob. 59IPCh. 13 - Prob. 60IPCh. 13 - Prob. 61IPCh. 13 - Prob. 62IPCh. 13 - Prob. 63IPCh. 13 - Prob. 64IPCh. 13 - Prob. 65IPCh. 13 - Prob. 66IPCh. 13 - Prob. 69IPCh. 13 - Prob. 70IPCh. 13 - Prob. 71IPCh. 13 - Prob. 72IPCh. 13 - Prob. 73IPCh. 13 - Prob. 74IPCh. 13 - Prob. 77CPCh. 13 - Prob. 79CPCh. 13 - Prob. 80CP
Knowledge Booster
Similar questions
- Show the products from reaction of p-bromoaniline with the following reagents: (a) CH3I (excess) (b) HCl (c) HNO2, H2SO4 (d) CH3COCl (e) CH3MgBr (f) CH3CH2Cl, AlCl3 (g) Product of (c) with CuCl, HCl (h) Product of (d) with CH3CH2Cl, AlCl3arrow_forwardTreatment of an aldehyde or ketone with cyanide ion (-:C=N), followed by protonation of the tetrahedral alkoxide ion intermediate, gives a cyanohydrin. Show the structure of the cyanohydrin obtained from cyclohexanone.arrow_forwardPropose a plausible mechanism for the following transformation: 1) Excess MeMgBr 2) H₂O HOarrow_forward
- 3. Provide a mechanism for the following transformation: Ph CI (2 eq.) 2 eq. NaCN 4. Provide a mechanism for the following transformation: S H₂O NC CN Ph Ph OH of OHarrow_forwardDraw a plausible mechanism for the following transformation: (a) N H₂O+ HO H + HH H₂C- CH3 1arrow_forwardIdentify products A and B from the given 1H NMR data. Treatment of acetone [(CH3)2C=O] with dilute aqueous base forms B. Compound B exhibits four singlets in its 1H NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1H) ppm. What is the structure of B?arrow_forward
- The last step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involves treatment of C with CH3Li to form an intermediate X, which forms β-vetivone with aqueous acid. Identify the structure of X and draw a mechanism for converting X to β-vetivone.arrow_forward3. Provide a mechanism for the following transformation: .OH H3C CH3 SOCI₂ CN + SO2 + 2 H3C 2 eq. pyridine H3C 4. Provide a mechanism for the following transformation: H + es-CEN DMF A -CENarrow_forwardPropose mechanisms for the following reactions. (b) OH H3PO4 CH₂OH heat H2SO4 heat (c) OH H2SO4 heat (d) OH H2SO4 heat CH2 CH3 8.5.0 + + +arrow_forward
- Identify products A and B from the given 1H NMR data.a. Treatment of CH2 = CHCOCH3 with one equivalent of HCl forms compound A. A exhibits the following absorptions in its 1H NMR spectrum: 2.2 (singlet, 3 H), 3.05 (triplet, 2 H), and 3.6 (triplet, 2 H) ppm. What is the structure of A?b. Treatment of acetone [(CH3)2C = O] with dilute aqueous base forms B. Compound B exhibits four singlets in its 1H NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1 H) ppm. What is the structure of B?arrow_forwardA) Propose an efficient synthesis for the following transformation: + en B) Propose an efficient synthesis for the following transformation: он + enarrow_forward(b) The Wieland Miescher ketone has been used in the synthesis of 50 synthetic and natural steroids. Propose a reasonable and stepwise reaction mechanism for the reaction formation of the Wieland Miescher ketone ion below: CH₂ CH₂ KOH Catalyst CH₁ H₂Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning