
Interpretation:
The structure of a given molecular formula C11H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C-13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
To Identify:
The structure of an alcohol of given molecular formula C11H6.
Broadband-decoupled spectrum:
The spectrum shows seven signals whereas the given molecular formula also has seven carbon atoms. Thus all the seven carbons have chemically different electronic environments showing signals.
- The signal in the region of 150-220 ppm indicates the carbon atom of carbonyl group (C=O).
- The three signals in the region of 10-25 ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene or methine groups.
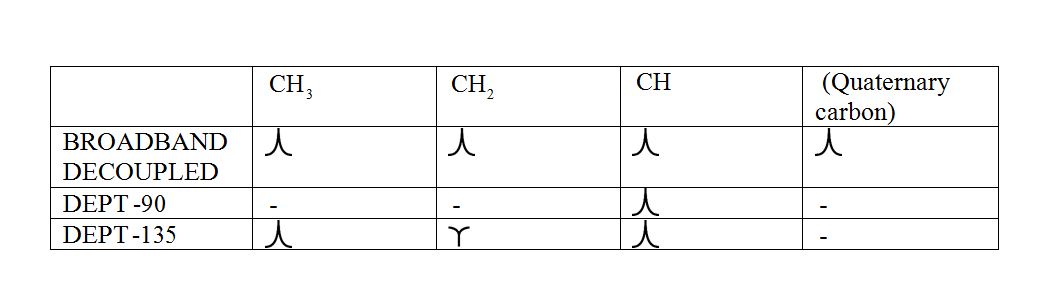
DEPT (Distortionless enhancement by polarization transfer):
a) DEPT-90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
b) DEPT-135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:
Trending nowThis is a popular solution!

Chapter 13 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Propose a structural formula for the analgesic phenacetin, molecular formula C10H13NO2, based on its 1H-NMR spectrum.arrow_forwardFollowing is the 1H-NMR spectrum of compound O, molecular formula C7H12. Compound O reacts with bromine in carbon tetrachloride to give a compound with the molecular formula C7H12Br2. The 13C-NMR spectrum of compound O shows signals at d 150.12, 106.43, 35.44, 28.36, and 26.36. Deduce the structural formula of compound O.arrow_forwardIndicate two basic differences that exist between the spectra of 1H y 13C in NMR.arrow_forward
- A compound with the molecular formula C7H14O exhibits the following 13C NMR spectra: Broadband-decoupled 220 200 180 160 140 120 100 80 -0 DEPT-90 220 200 180 160 140 120 100 80 DEPT-135 220 200 180 160 140 120 100 9-9 60 40 40 20 909 60 40 20 80 60 40 20 Chemical Shift (ppm) Several structures are consistent with these spectra. To determine which structure is correct, a ¹H NMR spectrum was acquired which exhibits five signals. One of those signals is a singlet at 1.9 ppm with an integration of 3, and another of the signals is a doublet at 0.9 ppm with an integration of 6. Using this information, draw the correct structure of the compound. Draw Your Solutionarrow_forwardShown below are carbon NMR spectra for various isomers of C5H11Br. Assign structures for the compound that would produce each spectrum.arrow_forwardThe 1H-NMR spectrum of compound R, C6H14O, consists of two signals: d 1.1 (doublet) and d 3.6 (septet) in the ratio 6:1. Propose a structural formula for compound R consistent with this informationarrow_forward
- The mass spectrum of 1-ethyl-1-methylcyclohexane shows many fragments, with two in very large abundance. Kne appears af m/z=111 and the other appears at m/z=97. Identify the structure of each of these fragments.arrow_forwardTreatment of 2-methylpropanenitrile [(CH3)2CHCN] withCH3CH2CH2MgBr, followed by aqueous acid, affords compound V, whichhas molecular formula C7H14O. V has a strong absorption in its IRspectrum at 1713 cm−1, and gives the following 1H NMR data: 0.91(triplet, 3 H), 1.09 (doublet, 6 H), 1.6 (multiplet, 2 H), 2.43 (triplet, 2 H), and2.60 (septet, 1 H) ppm. What is the structure of V?arrow_forwardCompound A has molecular formula C8H18. It shows one singlet in the ¹H-NMR spectrum. Identify A and explain your reasoning. For the toolbar, press ALT+F10 (PC) or ALT+FN+F10 (Mac). BIUS Paragraph V Arial = = = = +8HE {} X² X₂ PR хо 23 V 10pt >¶ ¶< - ABC :3 v V ✓ Ev AV ¶T "Ω Θ I 用く x B Q ► 8. Earrow_forward
- 10.9 A compound of molecular formula C₂H₂ClO₂ has peaks at 55.8, 56.8, 112.9, 113.4, 116.2, 123.1, 149.5, and 153.9 in its proton- decoupled ¹3C NMR spectrum. Give the structure of the compound and assign as many signals as possible to the carbons in the structure.arrow_forward4 The following is a monosubstituted aromatic hydrocarbon with the formula C9H12. Give its structure and assign chemical shifts. 1000 500 250 "H NMR 60 MHz 400 300 200 100 O CPS 100 50 Integral = 5 Integral = 6 Integral = 1 C9H12 8.0 7.0 6.0 5.0 4.0 3.0 2.0 1.0 O PPMarrow_forwardThe mass spectrum of 1-ethyl-1-methylcyclohexane shows many fragments, with two in very large abundance. One appears at m/z = 111 and the other appears at m/z = 97. Determine the identity and structure of each of these fragmentsarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
