
Concept explainers
Interpretation:
Among the given options, the correct pairing of prefix and the numbering system used for locating substituents has to be chosen.
Concept Introduction:
Benzene derivatives are compounds that contain benzene as the core with substituents in the benzene ring. This is done by replacement of one or more hydrogen atoms with other atoms in the ring. Substituents like alkyl groups or halogen groups are generally found. IUPAC name for monosubstituted benzene derivative use the name of the substituents as prefix to the name benzene.
If two substituents are present in the benzene ring, then it is known as disubstituted benzene derivative. Three isomers are possible for disubstituted benzene. The three isomers can be given as,
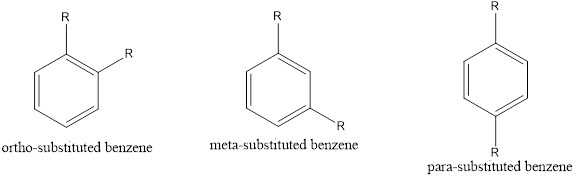
If the substitution is in first and second position, then it is known as ortho substitution. If the substitution is in first and third position, then it is known as meta substitution. If the substitution is in first and fourth position, then it is known as para substitution.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Give an example of each of the following: a. epimer b. acetal linkage c. reducing sugar d. monosaccharide e. anomer f. diastereomerarrow_forwardDefine the following terms: a. alkylating agents b. base analogue c. nonalkylating agent d. intercalating agent e. ethidium bromidearrow_forwardThe compounds 1-hexanol, 3-methyl-3-pentanol, hexanal, and 2-hexanone, and were used using different tests. Shown below is the schematic diagram of the tests performed on the compounds. Determine the identities of each compound based on the tests performed. a.) Compound A b.) Compound B c.) Compound C d.) Compound D Choices: I. 1-hexanol II. hexanal III. 2-hexanone IV. 3-methyl-3-pentanoarrow_forward
- Following are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses. Identify the pairs that are enantiomers. CHO сно H-C- OH H-C-OH H-C- OH но-с — н н-с—он но- ČHOH ČH,OH сно CHO Н-с—он но—с— н H-C- OH H-C-OH но—с—н Н-с—он ČHOH ČH,OH сно сно н-с—он но—с —н но—с— н но -с — н H-C- OH но- C-H ČH,OH ČH,OHarrow_forwardDraw the structure of the tripeptide alanylglycylvaline and determine its name using three-letter abbreviations.arrow_forwardDefine the following terms:a. isoprenoidb. isoprene unitc. terpened. carotenoide. mixed terpenoidarrow_forward
- Listed below are descriptions that may belong to amylose, amylopectin, both, or it may not belong to either. Write A if it describes amylose, B if it describes amylopectin, AB if the statement applies to both, or O if it doesn’t apply to either. 1. Its monosaccharides are bound by at least one α-1,4-glycosidic bond. 2. It contains α-1,6-glycosidic bonds. 3. It can be broken down by the enzyme α-amylase. 4. A polysaccharide made up of gulose. 5. It can form a double helix.arrow_forwardHow many stereoisomers are possible for D-xylulose? A) 10 B) 6 C) 8 D) 4arrow_forwardAssign a name to each of the following monosaccharides. Use D and L designations. H b. CH 2OH a. C=0 C=0 HO-C-H H-C-OH HO-C-H Н-С-Он CH,OH CH 2OH a. b. Circle all chiral centers for the monosaccharides shown. С. d. True or False: The molecules shown can rotate plane polarized light?arrow_forward
- Classify each of the following sugar pairs as enantiomers, diastereomers, epimers, or an aldose–ketose pair. a. D-erythrose and D-threose b. D-glucose and D-mannose c. D-ribose and L-ribose d. D-allose and D-galactose e. D-glyceraldehyde and dihydroxyacetonearrow_forwardwrite the introduction for the effect of denaturing agent on the structure of phycocyanin on two pagesarrow_forwardThe analgesic phenacetin is synthesized by treating 4- ethoxyaniline with acetic anhydride. a. Which of the following is the structure of 4- ethoxyaniline? OCH₂CH3 NH₂ OCH₂CH3 NH₂ OCH₂CH3arrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
