
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.7, Problem 13.5P
Following are two constitutional isomers with the molecular formula C4H8O2.
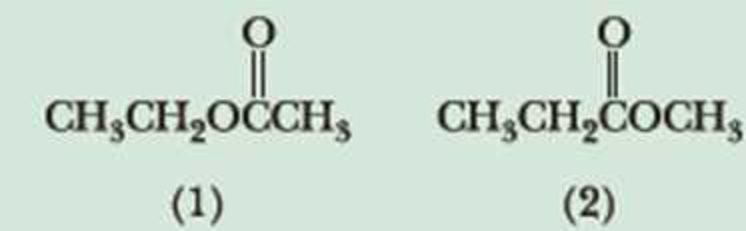
- (a) Predict the number of signals in the 1H-NMR spectrum of each isomer.
- (b) Predict the ratio of areas of the signals in each spectrum.
- (c) Show how you can distinguish between these isomers on the basis of chemical shift.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Logically deduce the structure of compound 1B whose Spectra are given:
Molecular formula C4H8O
Predict the number of signals in the 1H-NMR spectrum of each isomer.
Describe the 1H NMR spectrum of each compound. State how many NMR signals are present, the splitting pattern for each signal, and the approximate chemical shift
Chapter 13 Solutions
Organic Chemistry
Ch. 13.2 - Calculate the ratio of nuclei in the higher spin...Ch. 13.5 - State the number of sets of equivalent hydrogens...Ch. 13.5 - Each compound gives only one signal in its 1H-NMR...Ch. 13.6 - The line of integration of the two signals in the...Ch. 13.7 - Following are two constitutional isomers with the...Ch. 13.8 - Following are pairs of constitutional isomers....Ch. 13.10 - Following is a 1H-NMR spectrum of 2-butanol....Ch. 13.11 - Explain how to distinguish between the members of...Ch. 13 - Prob. 13.9PCh. 13 - Prob. 13.10P
Ch. 13 - Prob. 13.11PCh. 13 - Following are structural formulas for three...Ch. 13 - Following arc structural formulas for the cis...Ch. 13 - Prob. 13.14PCh. 13 - Following are three compounds with the molecular...Ch. 13 - Following are 1H-NMR spectra for compounds D, E,...Ch. 13 - Following are 1H-NMR spectra for compounds G, H,...Ch. 13 - Propose a structural formula for compound J,...Ch. 13 - Compound K, molecular formula C6H14O, readily...Ch. 13 - Compound M, molecular formula C5H10O, readily...Ch. 13 - Following is the 1H-NMR spectrum of compound O,...Ch. 13 - Treatment of compound P with BH3 followed by...Ch. 13 - The 1H-NMR spectrum of compound R, C6H14O,...Ch. 13 - Write structural formulas for the following...Ch. 13 - Prob. 13.25PCh. 13 - Ascaridole is a natural product that has been used...Ch. 13 - The 13C-NMR spectrum of 3-methyl-2-butanol shows...Ch. 13 - Prob. 13.28P
Additional Science Textbook Solutions
Find more solutions based on key concepts
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
During the early part of the 20th century, sulfanilamide (an antibacterial drug) was only administered by injec...
Elementary Principles of Chemical Processes, Binder Ready Version
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
Answer: _____
Organic Chemistry As a Second Language: Second Semester Topics
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
22.102 Write the structures of the cis and tram isomers, if any, for the following compounds:
Chemistry: The Molecular Nature of Matter
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The 1H NMR spectrum, 13C NMR spectrum, mass spectrum, and IR spectrum below belong to a chemical with the molecular formula C4H9XO, where X is a halogen. Provide a structure for that compound. You must explain how you determined the structure for full credit based on the data bellow.arrow_forwardIndicate how many different 1H-NMR signals are expected for the following molecules and describe their multiplicity (splitting).arrow_forwardWhich of the following compounds of molecular formula C6H1002, shows two singlets in its' H-NMR with areas of 2: 3, where the signal that integrates by 2 has a greater chemical shift?arrow_forward
- The 13C NMR spectrum of a compound with molecular formula C7H16O, measured in CDCl3. Assign the carbons by putting the appropriate letter over each peak in the spectrumarrow_forwardIII) Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. Predict the 1H NMR spectrum for each compound (include integration, multiplicity, and approximate chemical shift). For each set, be sure that you put them in a data table format!arrow_forwardWhat is the 1H NMR data (chemical shift, integration, multiplicity (specify as singlet, doublet, triplet,etc.) of the following two compounds?arrow_forward
- How would integration distinguish the 1H NMR spectra of the following compounds?arrow_forwardDetermine the multiplicity and predict the chemical shifts of each signal in the expected 1H NMR spectrum of the following compound.arrow_forwardThe Newman projection of an alkane with the molecular formula C8H18 is shown in the box. Predict the number of signals in 13C NMR. (please explain in simple terms)arrow_forward
- V.) A compound with molecular formula C8H10O has the following proton NMR spectrum. Determine the number of protons giving rise to each signal (a, b, c, and d). (SEE PICTURE ATTACHED).arrow_forwardDescribe the 1H-NMR spectrum of 4-bromoaniline (chemical shift, integration, multiple). Make a table and describe the results.arrow_forward07) How could 1H and 13C NMR distinguish between substances in each of the following pairs?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY