
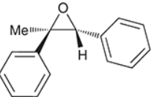
(a)
Interpretation: A starting material and reagents used to form following epoxide needs to be identified.
Concept introduction:
Preparation of epoxide-
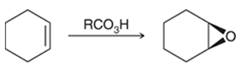
Peroxy acids generally used in this process are MCPBA and peroxyacetic acid. Formation of epoxide via peroxy acid us a stereospecific process thus, cis substituents in alkene (starting material) remain at cis to each other in the epoxide (product). Similarly, trans substituents in alkene remain at trans to each other.
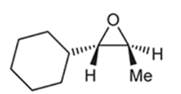
(b)
Interpretation: A starting material and reagents used to form following epoxide needs to be identified.
Concept introduction:
Preparation of epoxide- Epoxides can be prepared using peroxy acids. Alkenes are starting material in the preparation of epoxides with peroxy acids. Here, general reaction is represented as follows:
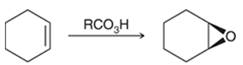
Peroxy acids generally used in this process are MCPBA and peroxyacetic acid. Formation of epoxide via peroxy acid us a stereospecific process thus, cis substituents in alkene (starting material) remain at cis to each other in the epoxide (product). Similarly, trans substituents in alkene remain at trans to each other.
(c)
Interpretation: A starting material and reagents used to form following epoxide needs to be identified.
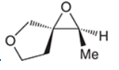
Concept introduction:
Preparation of epoxide- Epoxides can be prepared using peroxy acids. Alkenes are starting material in the preparation of epoxides with peroxy acids. Here, general reaction is represented as follows:
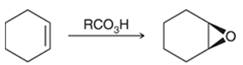
Peroxy acids generally used in this process are MCPBA and peroxyacetic acid. Formation of epoxide via peroxy acid us a stereospecific process thus, cis substituents in alkene (starting material) remain at cis to each other in the epoxide (product). Similarly, trans substituents in alkene remain at trans to each other.
(d)
Interpretation: A starting material and reagents used to form following epoxide needs to be identified.
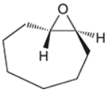
Concept introduction:
Preparation of epoxide- Epoxides can be prepared using peroxy acids. Alkenes are starting material in the preparation of epoxides with peroxy acids. Here, general reaction is represented as follows:
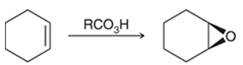
Peroxy acids generally used in this process are MCPBA and peroxyacetic acid. Formation of epoxide via peroxy acid us a stereospecific process thus, cis substituents in alkene (starting material) remain at cis to each other in the epoxide (product). Similarly, trans substituents in alkene remain at trans to each other.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
ORGANIC CHEMISTRY (LL) >CUSTOM PACKAGE<
- Rank the following carbonyl compounds in order of increasing reactivity in nucleophilic substitution reactions: H I II III IV ZIarrow_forwardDraw the major organic product formed in each of the following transition-metal catalyzed reactions:arrow_forwardPredict the expected product for each reaction and provide IUPAC name for the correct starting material to yield the desired epoxide:arrow_forward
- Provide the reagents needed to accomplish the following transformationsarrow_forwardProvide the reagents necessary to accomplish each of the following transformation.arrow_forwardDraw the products obtained in each of the following reactions, assuming that only one equivalent of each reagent is used in each case:arrow_forward
