
Concept explainers
Describe the
the splitting pattern for each signal, and the approximate chemical shift?
a. 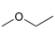 b.
b. 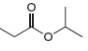 c.
c. 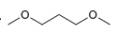 d.
d. 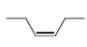
(a)
Interpretation: The
Concept introduction: The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.18P
The
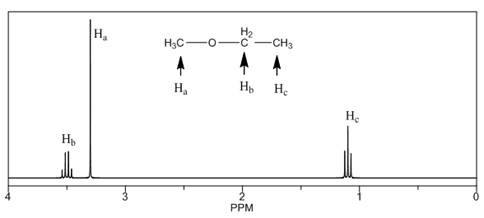
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is ethyl methyl ether
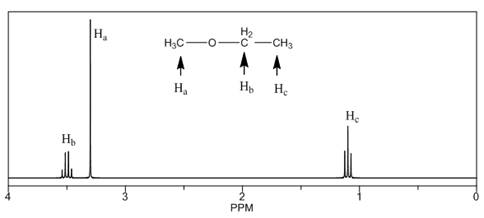
Figure 1
The proton
The
(b)
Interpretation:The
Concept introduction:The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.18P
The
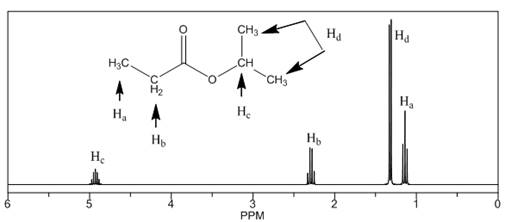
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is isopropylpropionate
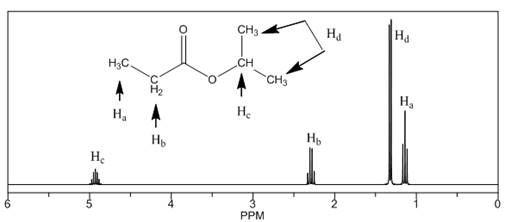
Figure 2
The proton
The
(c)
Interpretation: The
Concept introduction: The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.18P
The
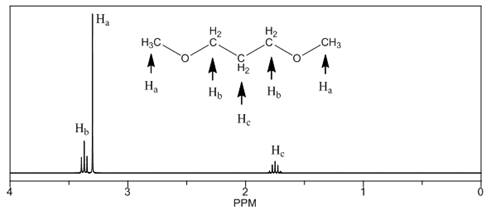
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is
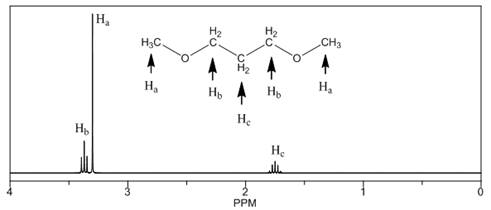
Figure 3
The proton
The
(d)
Interpretation: The
Concept introduction: The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.18P
The
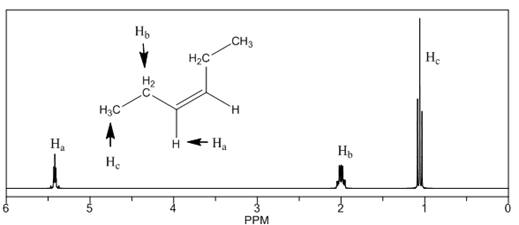
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is

Figure 4
The proton
The
Want to see more full solutions like this?
Chapter 14 Solutions
ORGANIC CHEM.(LL)W/STD GDE.+CONNECT PKG
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
Chemistry: A Molecular Approach (4th Edition)
Chemistry by OpenStax (2015-05-04)
Principles of General, Organic, Biological Chemistry
Thermodynamics, Statistical Thermodynamics, & Kinetics
- Consider geraniol, the principal constituent of rose oil.a.How many 1H NMR signals does geraniol exhibit? b.How many 13C NMR signals does geraniol exhibit? c.Into how many peaks will the protons on each C=C be split?arrow_forwardConsider geraniol, the principal constituent of rose oil. a.) How many 1H NMR signals does geraniol exhibit? b.) How many 13C NMR signals does geraniol exhibit?c.) Into how many peaks will the protons on each C=C be split?arrow_forwardExplain why the answer is A regarding to H-NMR belowarrow_forward
- Answer the following questions about each of the hydroxy ketones: 1-hydroxybutan-2-one (A) and 4-hydroxybutan-2-one (B). a.) How many signals are observed in the 1H NMR spectrum?b.) Give the splitting observed for each type of proton as well as its approximate chemical shift.arrow_forwardDraw an isomer of dichlorocyclopropane that gives an 1H NMR spectrum a. with one signal. b. with two signals. c. with three signals.arrow_forwardI. Using spectrum A (H-NMR) of the given compound, draw the corresponding COSY 2D-NMR Spectrum. II. Using spectrum A (H-NMR) and spectrum B (C-NMR) of the given compound, draw the corresponding HETCOR 2D-NMR Spectrum.arrow_forward
