
Concept explainers
(a)
Interpretation:
The product on reaction of
Concept introduction:
The
Answer to Problem 14.26AP
The product on reaction of
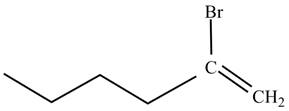
Explanation of Solution
The reaction of
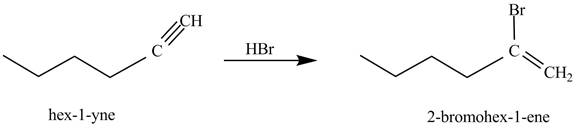
Figure 1
In the above reaction, hexyne reacts with ![]() to form a haloalkene. Therefore, the product on reaction of
to form a haloalkene. Therefore, the product on reaction of
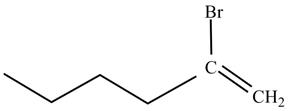
Figure 2
The product on reaction of
(b)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
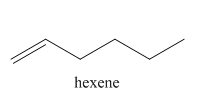
Explanation of Solution
The reaction of
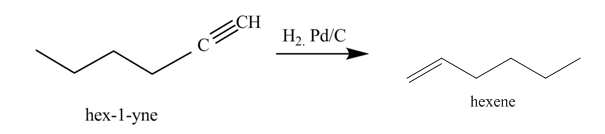
Figure 3
In the above reaction, hexyne reacts with hydrogen to form hexene. Hexyne is an unsaturated molecule consisting of a triple bond. It reacts hydrogen in presence of

Figure 4
The product on reaction of
(c)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
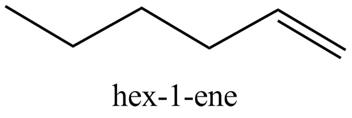
Explanation of Solution
The reaction of
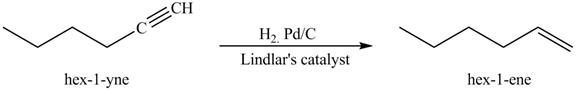
Figure 5
In the above reaction, hexyne reacts with
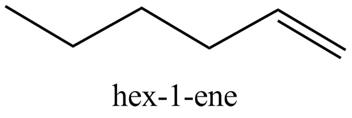
Figure 6
The product on reaction of
(d)
Interpretation:
The product on reaction of the product formed in part (c) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (c) with
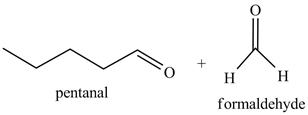
Explanation of Solution
The product formed in part (c) is shown below.
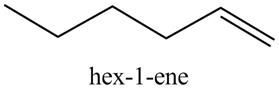
Figure 7
The reaction of the product formed in part (c) with
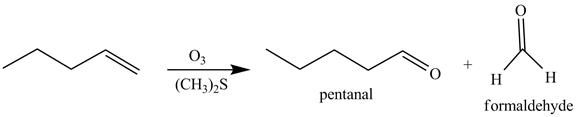
Figure 8
The above reaction is known as ozonlysis reaction. In the above reaction, a double bond is cleaved and oxidized to give two products. Reaction of hexene with

Figure 9
The products on reaction of reaction of the product formed in part (c) with
(e)
Interpretation:
The product on reaction of the product formed in part (c) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (c) with
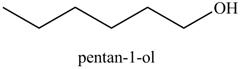
Explanation of Solution
The product formed in part (c) is shown below.

Figure 7
The reaction of above compound with
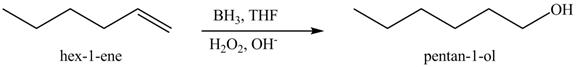
Figure 10
In the above reaction, hexene reacts with boron hydride to form organoborane which further reacts with peroxide to form an alcohol. The alcohol thus formed is by anti-markovnikov addition.

Figure 11
The product on reaction of the product formed in part (c) with
(f)
Interpretation:
The product on reaction of the product formed in part (c) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (c) with
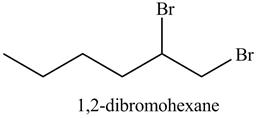
Explanation of Solution
The product formed in part (c) is shown below.
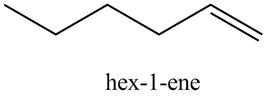
Figure 7
The reaction of above compound with
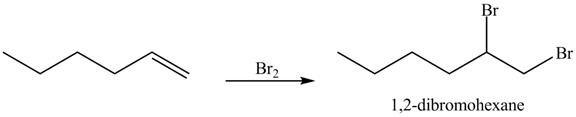
Figure 12
In the above reaction, hexene being unsaturated reacts with bromine molecule to form a dibromoalkane, that is
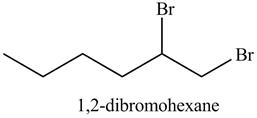
Figure 13
The product on reaction of the product formed in part (c) with
(g)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
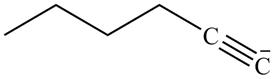
Explanation of Solution
The reaction of
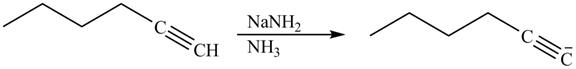
Figure 14
Alkynes react with sodamide

Figure 15
The product on reaction of
(h)
Interpretation:
The product on reaction of the product formed in part (g) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (g) with
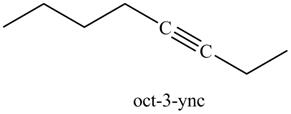
Explanation of Solution
The product formed in part (g) is shown below.
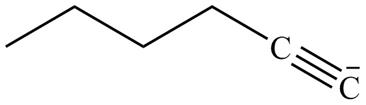
Figure 15
The reaction of above compound with
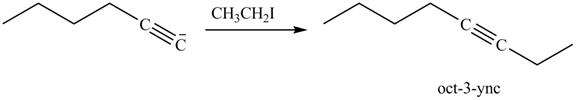
Figure 16
In the above reaction, the anion of alkyne obtained reacts with iodoethane to form an alkyne of eight carbons. It is non-terminal alkyne, that is, triple bond is not situated at the terminal end. The product formed is shown below.
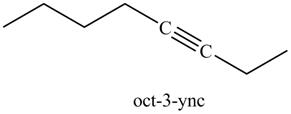
Figure 17
The product on reaction of the product formed in part (g) with
(i)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
Explanation of Solution
The reaction of
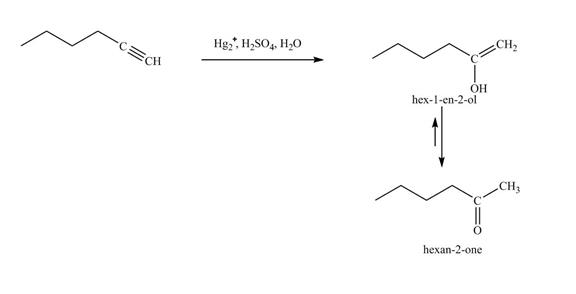
Figure 18
In the above reaction, hexyne reacts with

Figure 19
The product on reaction of
(j)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
Explanation of Solution
The reaction of
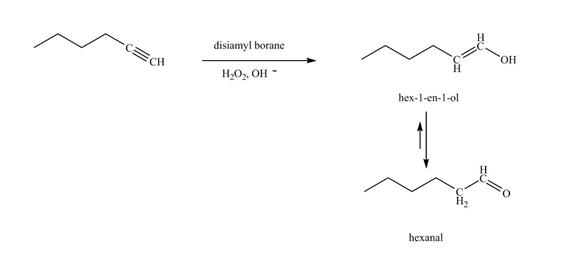
Figure 20
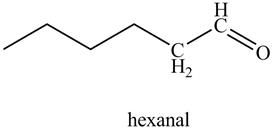
In the above reaction, hexyne reacts with disiamyl borane to form an organoborane. This reaction is also known as hydroboration-oxidation reaction. The organo-borane gives an enol which tautomerism to form an
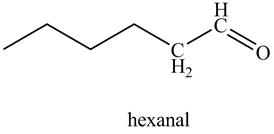
Figure 21
The product on reaction of
(k)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The products formed on reaction of
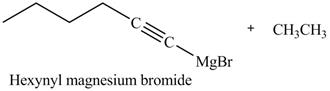
Explanation of Solution
The reaction of
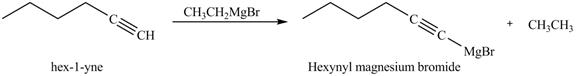
Figure 22
In the above reaction,

Figure 23
The products formed on reaction of
(l)
Interpretation:
The product on reaction of the product formed in part (k) with ethylene oxide and
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (k) with ethylene oxide and
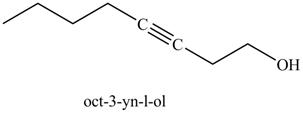
Explanation of Solution
The product formed in part (k) is shown below.
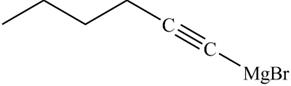
Figure 23
The reaction of above compound with ethylene oxide and
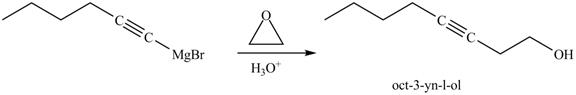
Figure 24
In the above reaction, the Grignard reagent obtained in part (k) reacts with ethylene oxide followed by hydrolysis to form an alcohol with a triple bond in between the chain. The product formed on reaction of product formed in part (k) with ethylene oxide and
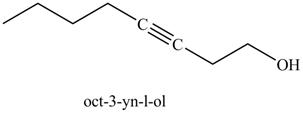
Figure 25
The product on reaction of the product formed in part (k) with ethylene oxide and
Want to see more full solutions like this?
Chapter 14 Solutions
Loose-leaf Version For Organic Chemistry
- Hydrocarbon X has the formula C6H12.X reacts with one molar equivalent of hydrogen in the presence of a palladium catalyst to form a product having 12 primary hydrogens.Treatment of X with ozone followed by zinc in aqueous acid gives a mixture two aldehydes.What is the structure of X?arrow_forward2-bromo-2-methylbutane undergoes hydrolysis reaction with water, H2O toform compound W. Compound X and compound Y are produced when 2-bromo-2-methylbutane undergoes elimination reaction with alcoholic ofsodium hydroxide, NaOH. (i) Draw the structural formula of compounds W, X and Yarrow_forwardAmines are converted into alkenes by a two-step process called Hofmann elimination. SN2 reaction of the amine with an excess of CH3I in the first step yields an intermediate that undergoes E2 reaction when treated with silver oxide as base. Pentylamine, for example, yields 1-pentene. Propose a structure for the intermediate, and explain why it readily undergoes elimination.arrow_forward
- Compound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct. Can be more than one answerarrow_forwardAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.arrow_forwardHydrocarbon A, C9H12 absorbs 3 equiv. of hydrogen to give B, C9H18 when hydrogenated over a Pd/C catalyst. Treatment of A with aqueous H2SO4 and Hg(II) gives two isomeric ketones, C & D. Oxidation of A with KMnO4 gives acetic acid and the tricarboxylic acid E shown below. Propose a structure for A.arrow_forward
- (10pts) Compound A, C10H16, was found to be optically active. On catalytic reduction over a palladium catalyst, 2 equivalents of hydrogen were absorbed, yielding compound B, CioH2o. On ozonolysis of A, two fragments were obtained. One fragment was identified as acetic acid (CHCOOH). The other fragment, compound C, was an optically active carboxylic acid, C8H14O2. Write reactions, and draw the correct structures for A-C, explain your answer in detail.arrow_forwardA synthetic organic molecule, G, which contains both aldehyde and ether functional groups, is subjected to a series of reactions in a multi-step synthesis pathway. In the first step, G undergoes a Wittig reaction, leading to the formation of an alkene, H. Subsequently, H is treated with an ozone (O3) reagent followed by a reducing agent in an ozonolysis reaction, resulting in the formation of two different products, I and J. Considering the functional groups present in G and the nature of the reactions involved, what are the most probable structures or functional groups present in products I and J? A. I contains a carboxylic acid group, and J contains an aldehyde group. B. I contains a ketone group, and J contains an alcohol group. C. I and J both contain aldehyde groups. D. I contains an ester group, and J contains a ketone group. Don't use chat gpt.arrow_forwardCompound X is optically inactive and has the formula C 16H 16Br 2. On treatment with strong base, X gives hydrocarbon Y, C 16H 14. Compound Y absorbs 2 equivalents of hydrogen when reduced over a palladium catalyst and reacts with ozone to give two fragments. One fragment Z, is an aldehyde with formula C 7H 6O. The other fragment is glyoxal, (CHO)2. Which of the following answers is correct? Select all that are correct.arrow_forward
- Three constitutional isomers of molecular formula C 5H 8O can be converted to 1-pentanol (CH 3CH 2CH 2CH 2CH 2OH) on treatment with two equivalents of H 2 in the presence of a Pd catalyst. Draw the structures of the three possible compounds, all of which contain a carbonyl grouparrow_forwardStarting with acetylene and ethylene oxide as the only sources of carbon atoms, show how to prepare the compound Q.)1,6-Hexanediolarrow_forwardThree products with the molecular formula C6 H4BrCl form when bromobenzene is treated with chlorine, Cl2, in the presence of FeCl3 as a catalyst. Name and draw a structural formula for each product.arrow_forward

 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


