
Concept explainers
(a)
Interpretation:
The product on reaction of
Concept introduction:
The
Answer to Problem 14.27AP
The product on reaction of
Explanation of Solution
The reaction of
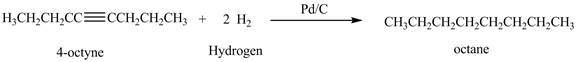
Figure 1
In the above reaction, octyne reacts with two moles of hydrogen to form octane. Octyne is an unsaturated molecule consisting of a triple bond. It reacts with hydrogen to form octane. Therefore, the product on reaction of
The product on reaction of
(b)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.27AP
The product on reaction of
Explanation of Solution
The reaction of

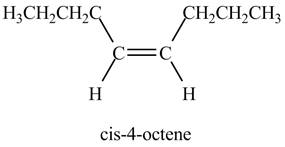
Figure 2
In the above reaction, octyne reacts with hydrogen to form octene. Octyne is an unsaturated molecule consisting of a triple bond. It reacts with hydrogen in presence of Lindlar’s catalyst to form
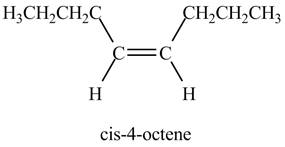
Figure 3
The product on reaction of
(c)
Interpretation:
The product on reaction of the product formed in part (b) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.27AP
The product on reaction of the product formed in part (b) with
Explanation of Solution
The product formed in part (b) is shown below.

Figure 4
The reaction of the product formed in part (b) with
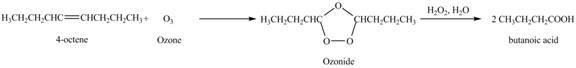
Figure 5
In the above reaction, octene a product from part (b) reacts with ozone to form ozonide. This ozonide formed hydrolysis to give two moles of butanoic acid as shown in figure 6. Therefore, the product formed on ozonolysis of octene is butanoic acid,
The product on reaction of the product formed in part (b) with
(d)
Interpretation:
The product on reaction of the
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.27AP
The product on reaction of the
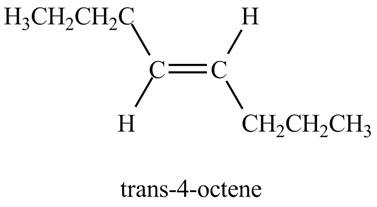
Explanation of Solution
The reaction of
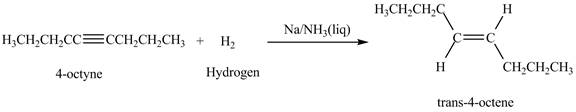
Figure 6
In the above reaction, octyne reacts with sodium in liquid ammonia to form

Figure 7
The product on reaction of the
(e)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.27AP
The product on reaction of the
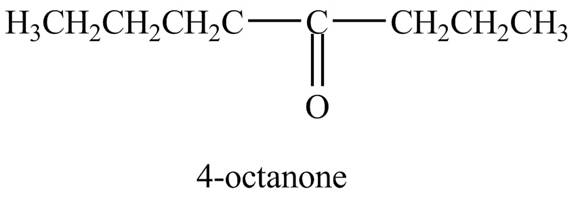
Explanation of Solution
The reaction of

Figure 8
In the above reaction, octyne reacts with
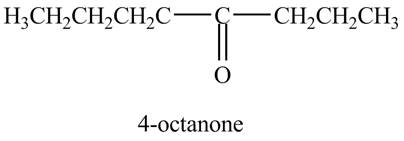
Figure 9
The product on reaction of the
(f)
Interpretation:
The product on reaction of the
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.27AP
The product formed on reaction of
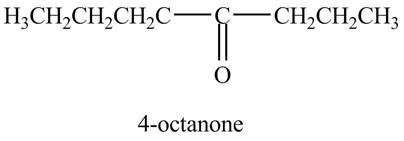
Explanation of Solution
The reaction of

Figure 10
In the above reaction, octyne reacts with boron hydride to form organoborane. Organoborane reacts with peroxide and hydroxide ion to form an enol. This enol tautomerises to form
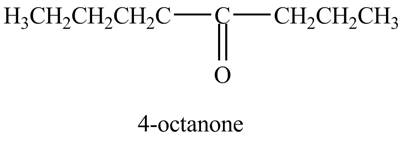
Figure 11
The product on reaction of the
Want to see more full solutions like this?
Chapter 14 Solutions
Organic Chemistry Study Guide and Solutions
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardUsing the data in Appendix C, determine which of the following bases is strong enough to deprotonate acetonitrile (CH3CN), so that equilibrium favors the products: (a) NaH; (b) Na2CO3; (c) NaOH; (d) NaNH2; (e) NaHCO3.arrow_forwarda) If the pH value of an aqueous solution of trimethylamine [(CH3)3N] is 10.75, what should be the molarity of this solution? (CH3)3N + H2O ↔ (CH3)3NH+ + OH-, Kb = 6,3 × 10-5 b) What will be the pH of the solution prepared by dissolving 8.35 g of aniline hydrochloride (C6H5NH3+Cl-) in 750 mL of 0.215 M aniline (C6H5NH2)? Is this solution an effective buffer? Explain (Kb = 7,4 × 10-10 for aniline, C: 12.0 g / mol, H: 1.0 g / mol, N: 14.0 g / mol, Cl: 35.4 g / mol) .arrow_forward
- Draw Lewis structures for the following compounds, andpredict which member of each pair will form hydrogen bonds:(a) NF₃ or NH₃(b) CH₃OCH₃ or CH₃CH₂OHarrow_forwarda) If the pH value of an aqueous solution of trimethylamine [(CH3) 3N] is 10.75, what should be the molarity of this solution? (CH3) 3N + H2O ↔ (CH3) 3NH + + OH-, Kb = 6.3 × 10-5 b) What will be the pH of the solution prepared by dissolving 8.35 g of aniline hydrochloride (C6H5NH3 + Cl-) in 750 mL of 0.215 M aniline (C6H5NH2)? Is this solution an effective buffer? Explain (Kb = 7.4 × 10-10 for aniline, C: 12.0 g / mol, H: 1.0 g / mol, N: 14.0 g / mol, Cl: 35.4 g / mol) .arrow_forwardIn a basic aqueous solution, esters react with hydroxide ion to form the salt of the carboxylic acid and the alcohol from which the ester is constituted. Name each of the following esters, and indicate the products of their reaction with aqueous base. (a) -OCH,CH3 (b) CH;CH2CH2-arrow_forward
- Write the reagent or draw structures of the starting material or organic product(s) in the following reactions. If more than one product is formed, identify the major product where possible. (a) (b) HO OH OH H2SO4 ? Cl₂ ? FeCl3arrow_forwardSuggest a possible structure for Compound X.arrow_forward(a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forward
- What is the product of this reaction? (A) OEt 1. LiAlH4 OEt 2.H+ (B) H ??? (C) OH (D) OHarrow_forwardIn an advanced synthetic chemistry experiment, a researcher prepares a compound, ZY-7, by reacting a ketone (C5H100) with hydroxylamine (NH2OH), followed by heating in the presence of an acid catalyst. The resulting compound, ZY-7, is then treated with a solution of sodium nitrite (NaNO2) and hydrochloric acid (HCI) at low temperature. Identify the class of compound that ZY-7 most likely belongs to after this series of reactions." A) Amide B) Oxime C) Nitro compound D) Diazonium salt E) Ester Don't use chatgpt please provide valuable answerarrow_forwardName and draw the products of each reaction. 1-propanol + hydrobromic acid → (a) (b) 1-pentanol H-SO A (c) N-propylmethanamide + water (d) 3-methyl oct-2-ene + hydrochloric acid -arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


