
Concept explainers
(a)
Interpretation:
The bond length of the indicated
Concept introduction:
Weak resonance contributors give some single-bond character to the two terminal
Answer to Problem 14.33P
In
Explanation of Solution
The bond length of
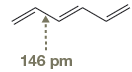
The normal
The amount of shortening of
(b)
Interpretation:
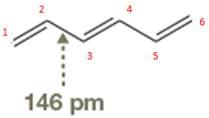
The bond length of the indicated
Concept introduction:
Resonance contributors give some double-bond character to the
Answer to Problem 14.33P
The occupied
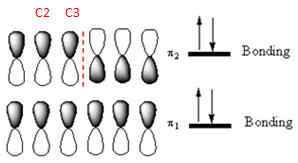
Explanation of Solution
The bond length of
The occupied
Out of these, the occupied

The occupied
(c)
Interpretation:
The bond length of the indicated
Concept introduction:
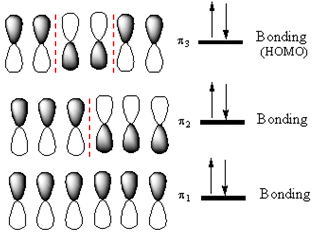
The highest energy MO of
Answer to Problem 14.33P
If an electron were promoted from the HOMO to the LUMO, the length of the
Explanation of Solution
The
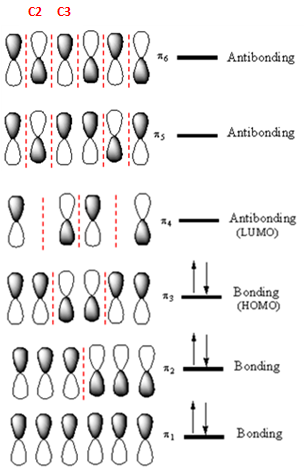
If an electron is promoted from HOMO (
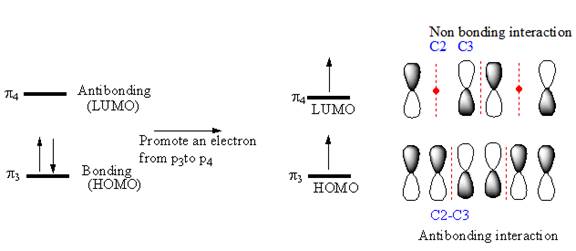
Therefore, the removal of an electron from the HOMO contributes to a shorter C2-C3 bond.
If an electron were promoted from the HOMO to the LUMO, the length of the
Want to see more full solutions like this?
Chapter 14 Solutions
ORGANIC CHEMISTRY:PRINCIPLES...(CL)
- The bond lengths of ethane, ethene, and ethyne were determined, and a graph of the C-H bond length vs. % s-character of the C hybrid orbital was made. Comment on the observed trend, additionally. How would this trend change if C-H bond energy vs. % s-character of the C hybrid orbital was graphed? Which hydrocarbon has the strongest carbon-carbon bond?arrow_forward4. Write a bond-line formula for the following compounds. a) HOOCCHCHCHCHCH2CCCH2CH2CH2COOH , (both double bonds in this compound have trans geometry)arrow_forwardThe wavelength at which the O2 molecule most stronglyabsorbs light is approximately 145 nm. (a) In which regionof the electromagnetic spectrum does this light fall?(b) Would a photon whose wavelength is 145 nm haveenough energy to photodissociate O2 whose bond energy is495 kJ/mol? Would it have enough energy to photoionize O2?arrow_forward
- In the methoxide anion (CH3O-), is it possible for a double bond to exist between C and O, given that the negative chargeresides on O? Explain why or why not.arrow_forward.For anion NO2-, (a) predict the stereochemistry. Justify your prediction with VSEPR theory and by formal charge; (b) indicate the hybridization appropriate to the stereochemistry; (c) describe the bond character of N-O for the resonance structure.arrow_forwardBecause the structure of a tetrahedrom molecule uses 3 dimensions, a tetrahedron has a bond angle of 109.5 instead of the expected 90 degrees. True or False?arrow_forward
- The cations O2+ and N2+ are formed when molecules of O2 and N2 are subjected to intense, high-energy solar radiation in Earths upper atmosphere. Write the electron configuration for O2+. Predict its bond order and magnetic behavior.arrow_forwardHow would I draw all the important resonance structures for cyanate (NCO1-)?arrow_forwardCalculate the bond order of the C2- Is its dissociation energy greater or smaller than that of C2? Is its bond length greater or smaller than that of C2?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


