
Concept explainers
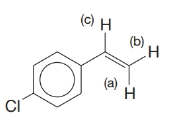
Given the following information, predict the appearance of the
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry: The Molecular Nature of Matter
Chemistry: The Central Science (13th Edition)
Chemistry: A Molecular Approach (4th Edition)
- The 1H NMR spectrum of chloromethane recorded on a 300 Hz NMR spectrometer consists of signals at 444 Hz and 1071 Hz downfield from TMS. Calculate the chemical shift of each absorption. Give answer in parts per million.arrow_forwardchromophores as (b) Identify the indicated NMR entiotopic/diastereotopic/homotopic magnetically equivalent or non-equivalent. as well as CH₂ H CH3 HOCHI HH₂C (c) A tri-substituted benzene possessing one bromine and two methoxy substituents exhibit three aromatic resonances at d = 6.40, 6.46, and 7.41 ppm. Identify the substitution pattern.arrow_forwardThe 1H NMR spectra of two carboxylic acids with molecular formula C3H5O2Cl are shown below. Identify the carboxylic acids. (The “offset” notation means that the farthest-left signal has been moved to the rightby the indicated amount to fit on the spectrum; thus, the signal at 9.8 ppm offset by 2.4 ppm has an actual chemical shift of 9.8 + 2.4 = 12.2 ppm.)arrow_forward
- The 'H NMR spectrum of 1,2-dimethoxyethane (CH;OCH,CH2OCH3) recorded on a 300 MHz NMR spectrometer consists of signals at 1017 Hz and 1065 Hz downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) At what frequency would each absorption occur if the spectrum were recorded on a 500 MHz NMR spectrometer?arrow_forwardDraw the structure for C5H10O with 1H NMR signals: δ 1.2 (s, 6H), 2.1 (bs, 1H), 5.0 (dd, 1H), 5.2 (dd, 1H), and 5.9 (dd, 1H).arrow_forwardThe 1H-NMR spectrum of 1,3-propanediol (HO-CH2-CH2-CH2-OH) shows a quintet at 1.81 ppm, a singlet at 2.75 pm, and a triplet at 3.83 ppm. Assign each signal to the protons it corresponds to in the molecule. Explain the splitting pattern observed for each signal.arrow_forward
- Cyclopentanone was treated with lithium aluminum hydride followed by H3O+. Explain what you would look for in the IR spectrum of the product to verify that the expected reaction had occurred. Identify which signal should be present and which signal should be absent.arrow_forward21arrow_forwardExplain the appearance of the 1H-NMR spectrum of 1,1,2-trichloroethane. How many signals would you expect, and into how many peaks will each of the signals be split?arrow_forward
- Deduce the structure of compound C.arrow_forwardWhat characteristics of the methyl benzoate spectrum rule out an aldehyde or carboxylicacid functional group giving the absorption at 1723 cm-1?arrow_forwardTreatment of 2-methylpropanenitrile [(CH3)2CHCN] withCH3CH2CH2MgBr, followed by aqueous acid, affords compound V, whichhas molecular formula C7H14O. V has a strong absorption in its IRspectrum at 1713 cm−1, and gives the following 1H NMR data: 0.91(triplet, 3 H), 1.09 (doublet, 6 H), 1.6 (multiplet, 2 H), 2.43 (triplet, 2 H), and2.60 (septet, 1 H) ppm. What is the structure of V?arrow_forward

 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



