
Conceptual Physical... -Masteringphysics
6th Edition
ISBN: 9780134079998
Author: Hewitt
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 50E
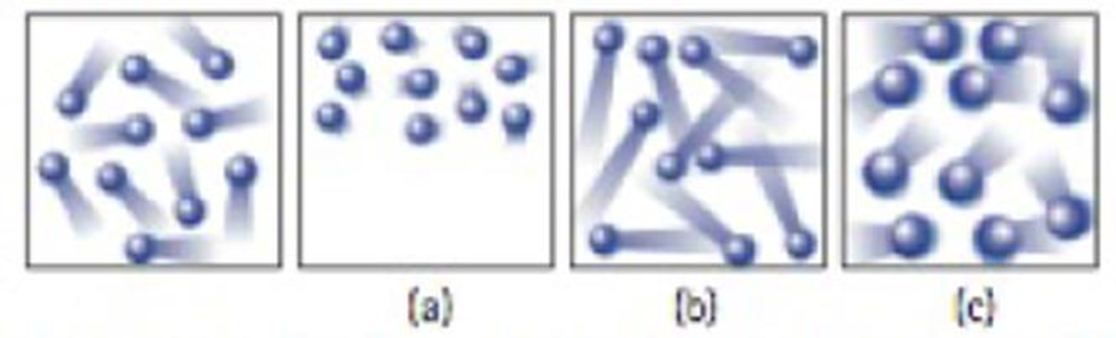
The leftmost diagram below shows the moving particles of a gas within a rigid container. Which of the three boxes on the right—(a), (b), or (c)—best represents this material upon the addition of heat?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Complete the table below to answer the question How do particles of solid, liquid and gas differ?
Given the table of coefficient (picture attached) of volume expansion above, which liquid expands the most? (use the volume expansion formula)
Please answer all the questions correctly and in details.
1. Describe the energy processes that occured.
2. State the new condition of the volume as the gas is heated.
3. Explain your answer in terms of motion the molecules.
4. Calculate the new volume
5. Calculate the average speed of the gas molecules at 95 degree celcius.
Chapter 14 Solutions
Conceptual Physical... -Masteringphysics
Ch. 14 - Prob. 1RCQCh. 14 - Prob. 2RCQCh. 14 - What pledge has been made by members of the...Ch. 14 - Prob. 4RCQCh. 14 - Prob. 5RCQCh. 14 - Prob. 6RCQCh. 14 - How are the particles in a solid arranged...Ch. 14 - How does the arrangement of particles in a gas...Ch. 14 - Which occupies the greatest volume: 1 g of ice, 1...Ch. 14 - What happens to the chemical identity of a...
Ch. 14 - What is a physical property?Ch. 14 - What doesnt change during a physical change?Ch. 14 - What is a chemical property.Ch. 14 - W hat is a chemical bond?Ch. 14 - What changes during a chemical reaction?Ch. 14 - Why is the freezing of water considered to be a...Ch. 14 - Prob. 17RCQCh. 14 - Why is the rusting of iron considered to be a...Ch. 14 - Prob. 19RCQCh. 14 - Prob. 20RCQCh. 14 - How many atoms are in a sulfur molecule that has...Ch. 14 - What is the difference between an element and a...Ch. 14 - How many atoms are there in one molecule of H3PO4?...Ch. 14 - What does the chemical formula of a substance tell...Ch. 14 - Prob. 25RCQCh. 14 - Prob. 26RCQCh. 14 - Prob. 27RCQCh. 14 - What is the chemical formula for the compound...Ch. 14 - Why are common names often used for chemical...Ch. 14 - How soon will nanotechnology give rise to...Ch. 14 - Prob. 31RCQCh. 14 - Who is the ultimate expert at nanotechnology?Ch. 14 - While visiting a foreign country, a...Ch. 14 - Of physics, chemistry, and biology, which science...Ch. 14 - Is chemistry the study of submicroscopic,...Ch. 14 - Prob. 47ECh. 14 - Prob. 48ECh. 14 - Which has stronger attractions among its...Ch. 14 - The leftmost diagram below shows the moving...Ch. 14 - The leftmost diagram below shows two phases of a...Ch. 14 - Prob. 52ECh. 14 - A skillet is lined with a thin layer of cooking...Ch. 14 - A cotton ball dipped in alcohol is wiped across a...Ch. 14 - Prob. 56ECh. 14 - Prob. 57ECh. 14 - Prob. 58ECh. 14 - Prob. 59ECh. 14 - Prob. 60ECh. 14 - Each night you measure your height just before...Ch. 14 - State whether each of the following is a physical...Ch. 14 - State whether each of the following is a physical...Ch. 14 - Classify each change as physical or chemical. Even...Ch. 14 - Classify each change as physical or chemical. Even...Ch. 14 - Prob. 66ECh. 14 - Is aging primarily an example of a physical or...Ch. 14 - Prob. 68ECh. 14 - Oxygen atoms are used to make water molecules....Ch. 14 - Oxygen, O2, is certainly good for you. Does it...Ch. 14 - Prob. 71ECh. 14 - If you eat metallic sodium or inhale chlorine gas,...Ch. 14 - Which of the following boxes contains only an...Ch. 14 - Prob. 74ECh. 14 - What is the chemical name for a compound with the...Ch. 14 - Prob. 76ECh. 14 - Prob. 77ECh. 14 - Prob. 78ECh. 14 - Prob. 79ECh. 14 - Prob. 80ECh. 14 - Medicines, such as pain relievers and...Ch. 14 - Your friend smells cinnamon coming from an...Ch. 14 - Chemistry is the study of (a) matter. (b)...Ch. 14 - The phase in which atoms and molecules no longer...Ch. 14 - Prob. 4RATCh. 14 - Based on the information given in the following...Ch. 14 - Prob. 6RATCh. 14 - Prob. 7RATCh. 14 - If you burn 50 g of wood and produce 10 g of ash,...Ch. 14 - If you have one molecule of TiO2, how many...Ch. 14 - Prob. 10RAT
Additional Science Textbook Solutions
Find more solutions based on key concepts
Does self-inductance depend on the value of the magnetic flux? Does depend on the current through the wire? Cor...
University Physics Volume 2
29.5 A circular loop of wire with a radius of 12.0 cm and oriented in the horizontal xy-plane is located in a r...
University Physics with Modern Physics (14th Edition)
What class of motion, natural or violent, did Aristotle attribute to motion of the Moon?
Conceptual Physics (12th Edition)
The height of a certain hill (in feet) is given by , where y is the distance (in miles) north, x the distance e...
Introduction to Electrodynamics
11. A block oscillating on a spring has period t = 2.0 s.
a. What is the period if the block's mass is doubled?...
College Physics: A Strategic Approach (4th Edition)
Explain all answers clearly, with complete sentences and proper essay structure, if needed. An asterisk (*) des...
The Cosmic Perspective Fundamentals (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Suppose you have one cubic meter of gold, two cubic meters of silver, and six cubic meters of aluminum. Rank them by mass, from smallest to largest, (a) gold, aluminum, silver (b) gold, silver, aluminum (c) aluminum, gold, silver (d) silver, aluminum, goldarrow_forward⦁ In a standard gas grill propane tank, there is approximately 4,579 mL of propane (C3H8). At a temperature of 55˚C, the tank has a pressure of 1,798 kPa. The tank is cooled to 25˚C and the pressure reduced to 1,025 kPa. What will the new volume be? Remember to pay close attention to the units of temperature before beginning your calculations.arrow_forwardPart 1. A slightly inflated balloon is placed about eight centimeters above a burning candle. Explain the change to the size of the balloon based on the kineticmolecular theory.Part 2. The same balloon is now placed in a bucket of ice cold water. Explain the change to the size of the balloon based on the kinetic molecular theory.In both cases, assume the balloon is tied tight enough so that air does not escape.arrow_forward
- Please Kindly Help me to answer this. Thank you very much! Note: Round off your final answer to the nearest ten-thousandth. Take note: Only the final answer should be rounded off. Do NOT write answers in scientific notation. Always write the answer with 4 decimal places, (i.e, 4.25 should be written as 4.2500) EXCEPT for whole numbers. Question: Steel railroad tracks are laid when the temperature is 5.0 °C. A standard section of rail is then 12.0 m long. The gap should be left between rail sections so that there is no compression when the temperature gets as high as 42 °C is ____________ cm.arrow_forwardDirections: Analyze the illustration and answer the given questions below In what phase is the substance at 250 K and 50 atm pressure? What phases will exist at 1 atm and 195.5 K? What is the significance of pressure 5.2 atm? Describe the changes that take place when a sample of solid CO2 is heated at a pressure of 1 atm, starting at a temperature of 195.5Karrow_forwardMark the correct answer 1. The heat transmission mechanism that prevails when it feels cold when the wind blows is a) thermal insulation effect of outerwear b) thermal radiation through the whole body c) forced convection through air d) conduction through air 2. When a body receives energy in the form of heat a) its internal energy increases b) you have to completely change the aggregation state c) the mass of the body increases according to Einstein's equation d) is hotterarrow_forward
- Thermodynamics. Describe the principles of the following instruments used for accuratemeasurement of temperature. In your answer, draw diagrams showing thesetup for each instrument, state the temperature range in which they are usefuland give one important advantage of each instrument.(a) Magnetic thermometer(b) Optical pyrometer(c) Vapor pressure thermometerarrow_forwardIdentify the following and write your answer in the blanks. ____________4. The freezing point is 32^o F and the boiling point is 212^o F. ___________________________5. The study of macroscopic properties of matter such as heat and temperature.arrow_forwardPlease answer question and just send me the paper solutions asap dont type the answer question and use the formula from 2nd picture ASAParrow_forward
- Solve the following problems systematically and show how you arrived at the final answer. 1. Calculate amount of heat that must be absorbed by 10.0 gram of ice at 200C to convert it to liquid water at 600C. Given: Specific heat of ice = 2.1 J/g0C Specific heat of water = 4.18 J/g0C Heat fus = 6.0 kJ/molarrow_forwardwater has the highest density in which phase 1.plasma 2.liquid 3. solid 4. gasarrow_forwardThe heat capacity of a solid at constant volume is given, according to the Debye model, assee figure where ΘD is the Debye temperature, N the number of atoms, and kB the Boltzmann constant. Show that this tends to a constant at high temperatures.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY