
In 1654 Otto von Guericke, inventor of the air pump, gave a demonstration before the noblemen of the Holy Roman Empire in which two teams of eight horses could not pull apart two evacuated brass hemispheres, (a) Assuming the hemispheres have (strong) thin walls, so that R in Fig. 14-29 may be considered both the inside and outside radius, show that the force
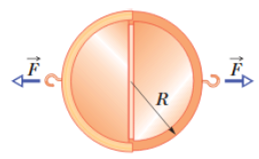
Figure 14-29 Problem 7.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Fundamentals of Physics, Volume 1, Chapter 1-20
Additional Science Textbook Solutions
An Introduction to Thermal Physics
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Mathematical Methods in the Physical Sciences
Conceptual Physics: The High School Physics Program
Applied Physics (11th Edition)
College Physics: A Strategic Approach (4th Edition)
- In 1923, the United States Army (there was no United States Air Force at that time) set a record for in-flight refueling of airplanes. Using two refueling planes, an Airco DH-4B biplane was able to remain in flight for 39 h. During the flight, the refueling planes were able to air-transfer a total of 687 gallons of fuel to the plane in 9 refueling transfers. Assume that the refueling nozzle had a diameter of 1.55 in and each refueling took 2.35 min to perform. Calculate the velocity of the fuel through the nozzle. Assume that the fuel filled the entire cross-sectional area of the nozzle.arrow_forwardIn 1923, the United States Army (there was no United States Air Force at that time) set a record for in-flight refueling of airplanes. Using two refueling planes, an Airco DH-4B biplane was able to remain in flight for 36 h. During the flight, the refueling planes were able to air-transfer a total of 687 gallons of fuel to the plane in 9 refueling transfers. Assume that the refueling nozzle had a diameter of 1.55 in and each refueling took 2.39 min to perform. Calculate the velocity of the fuel through the nozzle. Assume that the fuel filled the entire cross-sectional area of the nozzle. velocity: m/s TOOLS x10arrow_forwardIn 1923, the United States Army (there was no United States Air Force at that time) set a record for in-flight refueling of airplanes. Using two refueling planes, an Airco DH-4B biplane was able to remain in flight for 38 h. During the flight, the refueling planes were able to air-transfer a total of 687 gallons of fuel to the plane in 9 refueling transfers. Assume that the refueling nozzle had a diameter of 1.35 in and each refueling took 2.47 min to perform. Calculate the velocity of the fuel through the nozzle. Assume that the fuel filled the entire cross-sectional area of the nozzle. 22.82 velocity: m/s Incorrectarrow_forward
- Macmillan Learning In 1923, the United States Army (there was no United States Air Force at that time) set a record for in-flight refueling of airplanes. Using two refueling planes, an Airco DH-4B biplane was able to remain in flight for 35 h. During the flight, the refueling planes were able to air-transfer a total of 687 gallons of fuel to the plane in 9 refueling transfers. Assume that the refueling nozzle had a diameter of 1.65 in and each refueling took 2.35 min to perform. Calculate the velocity of the fuel through the nozzle. Assume that the fuel filled the entire cross-sectional area of the nozzle. velocity: m/sarrow_forwardA piece of aluminum (bulk modulus 7.1 x 10¹0 N/m²) is placed in a vacuum chamber where the air pressure is 0.378 x 105 Pa. The vacuum pump is then turned on and the pressure is further reduced to zero. Determine the fractional change AV/V in the volume of the aluminum.arrow_forwardssm A patient recovering from surgery is being given fluid intravenously. The fluid has a density of 1030 kg/m², and 9.5 × 10-4 m³ of it flows into the patient every six hours. Find the mass 55. flow rate in kg/s.arrow_forward
- You are on an interstellar mission from the Earth to the 8.7 light-years distant star Sirius. Your spaceship can travel with 70% the speed of light and has a cylindrical shape with a diameter of 6m at the front surface and a length of 25 m. You have to cross the interstellar medium with an approximated density of 1 hydrogen atom/m³. (a) Calculate the time it takes your spaceship to reach Sirius. (b) Determine the mass of interstellar gas that collides with your spaceship during the mission. Note: Use 1.673 × 10¬27 kg as proton mass.arrow_forwardApproximate the change in the atmospheric pressure when the altitude increases from z=6 km to z= 6.05 km using the formula P(z) = 1000 e From z = 6 km to z=6.05 km, the change in atmospheric pressure is approximately (Type an exact answer.) etarrow_forwardGold, which has a density of 19.32 g/cm3, is the most ductile metal and can be pressed into a thin leaf or drawn out into a long fiber. (a) If a sample of gold with a mass of 3.663 g, is pressed into a leaf of 2.187 um thickness, what is the area of the leaf? (b) If, instead, the gold is drawn out into a cylindrical fiber of radius 2.200 um, what is the length of the fiber? (a) Number i 1.429 Units m^2 (b) Number 72.56 Unitsarrow_forward
- Gold, which has a density of 19.32 g/cm3, is the most ductile metal and can be pressed into a thin leaf or drawn out into a long fiber. (a) If a sample of gold, with a mass of 27.63 g, is pressed into a leaf of 1.000 mm thickness, what is the area of the leaf? (b) If, instead, the gold is drawn out into a cylindrical fiber of radius 2.500 mm, what is the length of the fiber?arrow_forwardYou have scored a great internship with NASA, working on planning for an upcoming mission to Mars. The transfer orbit to Mars will last for several months and will require reclamation of the oxygen in the carbon dioxide exhaled by the crew. In one method of reclamation, 1.00 mol of carbon dioxide produces 1.00 mol of oxygen and 1.00 mol of methane as a byproduct. The methane is stored in a tank under pressure and is available to control the orientation of the spacecraft by controlled venting. A single astronaut exhales 1.09 kg of carbon dioxide each day. If the methanegenerated in the respiration recycling of three astronauts during one week of flight is stored in an originally empty 150-L tank at -45.0°C, what is the final pressure in the tank?arrow_forwardGold, which has a density of 19.32 g/cm³, is the most ductile metal and can be pressed into a thin leaf or drawn out into a long fiber. (a) If a sample of gold with a mass of 8.278 g, is pressed into a leaf of 1.604 µm thickness, what is the area of the leaf? (b) If, instead, the gold is drawn out into a cylindrical fiber of radius 2.400 um, what is the length of the fiber? (a) Number i 2671.25 (b) Number Units Units acre-feetarrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





