
Concept explainers
To pick the keyword from given list: That fits for compounds having same molecular formula but different structural formulas.
Answer to Problem AM
Explanation of Solution
Hydrocarbons which have same molecular formula but have different structural, chemical and physical properties are known as isomers.
The hydrocarbons which have different structural formulas but their molecular formula is same then these compounds are known as structural isomers. They have same molecular mass but their chemical properties are different. Constitutional isomers are drawn by altering the structural formula of a hydrocarbon.
For example, two constituional isomers of a hydrocarbon
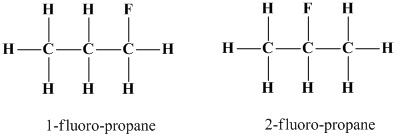
Conclusion:
Therefore, the word constitutional isomers can be picked from the given list.
Want to see more full solutions like this?
Chapter 14 Solutions
INTRO.TO PHYSICAL SCIENCE NSU PKG >IC<
- Use both full and condensed structural formulas to show the difference between methane and a methyl group and between ethane and an ethyl group.arrow_forwardIdentify each structural formula as belonging to an alkyl halide, alcohol, amine, carboxylic acid, ester, or amide. (a) CH3CH2NH2 (b) (c) (d) (e) (f)arrow_forwardWhat structural feature distinguishes an aromatic hydrocarbon from an aliphatic hydrocarbon?arrow_forward
- What two simpler sugars combine to form sucrose? Which sugar is the monomer of both starch and cellulose?arrow_forwardWhich of these structural formulas is valid, and which is incorrect? (a) (b)arrow_forwardGive the general molecular formulas for alkanes, cycloalkanes, alkenes, and alkynes. Write the structural formula for one example of each of these classes.arrow_forward
- State whether the structural formulas shown in each case represent the same compound, or are constitutional isomers, or are neither. (a) CH3CH2NH2 and CH3NHCH2CH3 (b) HOOCCH2CH3 and CH3CH2COOH (c)arrow_forwardGiven the IUPAC name, draw the structural formula for each alkyl halide. (a) 1,2,2-trifluorobutane (b) 1-bromo-2-chlorocyclopropanearrow_forwardAcrilan is an addition polymer made from the monomer named acrylonitrile (cyanoethene). Show by means of an equation how acrylonitrile polymerizes to Acrilan.arrow_forward
- Distinguish between saturated and unsaturated hydrocarbons.arrow_forwardWhich of the following is the most common aromatic compound? (14.2) (a) benzedrine (b) ethylene (c) butane (d) benzenearrow_forwardWhat characteristic group does an amine contain, and what are some applications for these compounds? What property makes the simple amines unpopular?arrow_forward
 An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
