
Concept explainers
a)
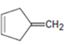
Interpretation:
Whether 4-methylenecyclopent-1-ene will have a π – π * UV adsorption in the 200 to 400nm range is to be stated.
Concept introduction:
Compounds having alternating double bonds (conjugated dienes) will have π – π *UV adsorption in the 200 to 400nm range.
To state:
Whether 4-methylenecyclopent-1-ene will have a π – π * UV adsorption in the 200 to 400nm range.
b)
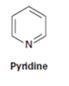
Interpretation:
Whether pyridine will have a π – π * UV adsorption in the 200 to 400nm range
Concept introduction:
Compounds having alternating double bonds (conjugated dienes) will have π – π * UV adsorption in the 200 to 400nm range.
To state:
Whether pyridine will have a π – π * UV adsorption in the 200 to 400nm range
c)
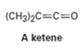
Interpretation:
Whether the ketene given will have a π – π * UV adsorption in the 200 to 400 nm range
Concept introduction:
Compounds having alternating double bonds (conjugated dienes) will have π – π * UV adsorption in the 200 to 400nm range.
To state:
Whether the ketene given will have a π – π * UV adsorption in the 200 to 400 nm range
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- What is the structure of the compound with the formula C6H12O2, if it has a strong IR signal near 2900 and 1740 cm-1, the 1H-NMR spectrum is: 1.12 ppm (3H triplet) 1.25 ppm (6H doublet) 2.27 ppm (2H quartet) 5.02 ppm (1H septet)arrow_forwardThe IR, and 1H NMR spectra of a compound with formula C5H6O2 are shown below. Determine the structure of the compound. You must fill all the parts to this question in the given spaces.arrow_forwardCompounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.arrow_forward
- An unknown compound has a molecular formula of C4H6O2. Its IR spectrum shows absorptions at 3095, 1762, 1254, and 1118 cm -1. It exhibits the following signals in its 1H NMR spectrum (ppm): 2.12 (singlet,3H), 4.55 (doublets of doublets, 1H), 4.85 (doublet of doublets, 1H), 7.25 (doublets of doublets, 1H); and the following signals in its 13C NMR spectrum (ppm): 20.8, 100.4, 141.2, 168.0. Draw the structure of the unknown compoundarrow_forwarddraw the strucutre based off the IR spectrum, 1H NMR spectrum, and 13C NMR spectrum for this compound. This compound is prepared from the anti-Markovnikov addition of water to phenylacetylene. Its molecular formula is C8H8O.arrow_forwardGiven the following compounds and boiling points: Determine the IMFs Order the compounds from weakest to strongest IMFs, and Pentane (C5H12) 36°C Methane (CH4) -164°C Sodium n-butoxide (C4H9NaO) +260°C Hydrogen dioxide (H2O) 100°Carrow_forward
- what is the compound that most easily reacts with bromine in addition? a) CH4 b) C2H2 c) C3H8 d) C4H10arrow_forwardIdentify the structures of D and E, isomers of molecular formula C6H12O2, from their IR and 1H NMR data. Signals at 1.35 and 1.60 ppm in the 1H NMR spectrum of D and 1.90 ppm in the 1H NMR spectrum of Eare multiplets.a. IR absorption for D at 1743 cm−1b. IR absorption for E at 1746 cm−1arrow_forwardAssign R or S designation to each of the following compounds. Questions I, II and III was already answered.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

