
(a)
The temperature and pressure at point
(a)
Answer to Problem 13P
The pressure at point
Explanation of Solution
There is
The cycle of the ideal gas is shown below in figure 1.
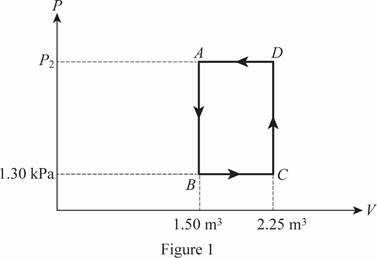
The pressure at point
Write the formula for the pressure at point
Here,
Write the formula for the temperature at point
Here,
Conclusion:
Substitute
Substitute
The pressure at point
(b)
The net work done on the gas if the gas completes 4 cycles.
(b)
Answer to Problem 13P
The net work done on the gas if the gas completes 4 cycles is
Explanation of Solution
There is
The cycle of the ideal gas is shown below in figure 1.
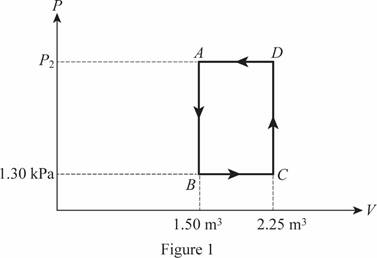
The pressure at point
Write the formula for the work done in four cycle of the gas.
Here,
Conclusion:
Substitute
The net work done on the gas if the gas completes 4 cycles is
(c)
The internal energy of the gas at point
(c)
Answer to Problem 13P
The internal energy of the gas at point
Explanation of Solution
There is
The cycle of the ideal gas is shown below in figure 1.

The pressure at point
Write the formula for the internal energy at point
Here,
Conclusion:
Substitute
The internal energy of the gas at point
(d)
The total change in internal energy of the gas during the four cycles.
(d)
Answer to Problem 13P
There is no change in internal energy of the gas through the complete four cycles.
Explanation of Solution
There is
The pressure at point
For ideal gases, change in internal energy depends on the change in temperature only. If the temperature remains same, there is no change in internal energy.
Since in the course of the complete four cycles of the gas, there is no change in temperature of the gas, there is no change in the internal energy.
Conclusion:
There is no change in internal energy of the gas through the complete four cycles.
Want to see more full solutions like this?
Chapter 15 Solutions
PHYSICS
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





