
Concept explainers
(a)
Interpretation:
To write the structural formula of the lowest molecular weight chiral molecule of
Concept Introduction:
A molecule is said to be chiral if it cannot be superimposed on its mirror image and if it does not possess an alternate axis of symmetry.
A carbon atom bonded in a tetrahedral structure to four different substituents in a molecule, it is called as a chiral centre or stereocentre.
If an organic molecule has more than one chiral carbon or chiral centre then the molecule may be achiral or chiral and it is depend upon whether the molecule has element of symmetry or not.
The elements of symmetry are-
- A plane of symmetry
- A centre of symmetry
- n-fold alternating axis of symmetry.
Answer to Problem 15.18P
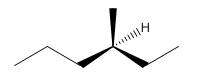
Explanation of Solution
Since alkanes consist of only carbon and hydrogen, the lowest molecular weight alkane can have four different groups attached to it namely- hydrogen, methyl group, ethyl group, and propyl group.
So the molecule so formed is: 3-Methyl-Hexane.
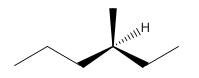
3-Methyl-Hexane.
(b)
Interpretation:
To write the structural formula of the lowest molecular weight chiral molecule of
Concept Introduction:
A molecule is said to be chiral if it cannot be superimposed on its mirror image and if it does not possess an alternate axis of symmetry.
A carbon atom bonded in a tetrahedral structure to four different substituents in a molecule, it is called as a chiral centre or stereocentre.
If an organic molecule has more than one chiral carbon or chiral centre then the molecule may be achiral or chiral and it is depend upon whether the molecule has element of symmetry or not.
The elements of symmetry are-
- A plane of symmetry
- A centre of symmetry
- n-fold alternating axis of symmetry.
Answer to Problem 15.18P
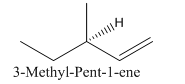
Explanation of Solution
Since alkenes consist of only carbon and hydrogen, with a double bond, the lowest molecular weight alkene can have four different groups attached to it namely- hydrogen, methyl group, vinyl group, and ethyl group.
So the molecule so formed is: 3-Methyl-Pent-1-ene.
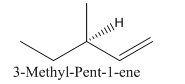
(c)
Interpretation:
To write the structural formula of the lowest molecular weight chiral molecule of alcohol.
Concept Introduction:
A molecule is said to be chiral if it cannot be superimposed on its mirror image and if it does not possess an alternate axis of symmetry.
A carbon atom bonded in a tetrahedral structure to four different substituents in a molecule, it is called as a chiral centre or stereocentre.
If an organic molecule has more than one chiral carbon or chiral centre then the molecule may be achiral or chiral and it is depend upon whether the molecule has element of symmetry or not.
The elements of symmetry are-
- A plane of symmetry
- A centre of symmetry
- n-fold alternating axis of symmetry.
Answer to Problem 15.18P
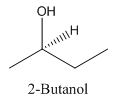
Explanation of Solution
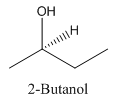
Since alcohol consist of carbon and hydrogen and oxygen, the lowest molecular weight alcohol can have four different groups attached to it namely- hydrogen, methyl group, hydroxyl group, and ethyl group.
So, the molecule so formed is: 2-Butanol.
(d)
Interpretation:
To write the structural formula of the lowest molecular weight chiral molecule of alcohol.
Concept Introduction:
A molecule is said to be chiral if it cannot be superimposed on its mirror image and if it does not possess an alternate axis of symmetry.
A carbon atom bonded in a tetrahedral structure to four different substituents in a molecule, it is called as a chiral centre or stereocentre.
If an organic molecule has more than one chiral carbon or chiral centre then the molecule may be achiral or chiral and it is depend upon whether the molecule has element of symmetry or not.
The elements of symmetry are-
- A plane of symmetry
- A centre of symmetry
- n-fold alternating axis of symmetry.
Answer to Problem 15.18P
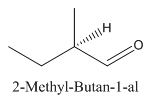
Explanation of Solution
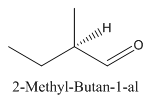
Since
So the molecule so formed is: 2-Methyl-Butan-1-al.
(e)
Interpretation:
To write the structural formula of the lowest molecular weight chiral molecule of alcohol.
Concept Introduction:
A molecule is said to be chiral if it cannot be superimposed on its mirror image and if it does not possess an alternate axis of symmetry.
A carbon atom bonded in a tetrahedral structure to four different substituents in a molecule, it is called as a chiral centre or stereocentre.
If an organic molecule has more than one chiral carbon or chiral centre then the molecule may be achiral or chiral and it is depend upon whether the molecule has element of symmetry or not.
The elements of symmetry are-
- A plane of symmetry
- A centre of symmetry
- n-fold alternating axis of symmetry.
Answer to Problem 15.18P
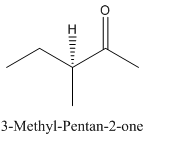
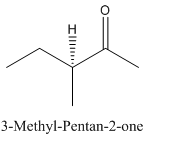
Explanation of Solution
Since
So, the molecule so formed is: 3-Methyl-Pentan-2-one.
(f)
Interpretation:
To write the structural formula of the lowest molecular weight chiral molecule of alcohol.
Concept Introduction:
A molecule is said to be chiral if it cannot be superimposed on its mirror image and if it does not possess an alternate axis of symmetry.
A carbon atom bonded in a tetrahedral structure to four different substituents in a molecule, it is called as a chiral centre or stereocentre.
If an organic molecule has more than one chiral carbon or chiral centre then the molecule may be achiral or chiral and it is depend upon whether the molecule has element of symmetry or not.
The elements of symmetry are-
- A plane of symmetry
- A centre of symmetry
- n-fold alternating axis of symmetry.
Answer to Problem 15.18P
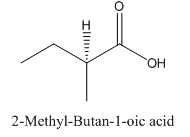
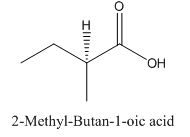
Explanation of Solution
Since carboxylic acid consist of carbon and hydrogen and oxygen, the lowest molecular weight carboxylic acid can have four different groups attached to it namely- hydrogen, methyl group, carboxylic acid group and ethyl group.
So, the molecule so formed is: 2-Methyl-Butan-1-oic acid.
Want to see more full solutions like this?
Chapter 15 Solutions
Bundle: Introduction To General, Organic And Biochemistry, 11th + Owlv2, 1 Term (6 Months) Printed Access Card
- 15-44 Consider the structure of the immunosuppressant FK-506, a molecule shown to disrupt calcineurin- mediated signal transduction in T-lymphocytes. What is the molecular formula of this immunosuppressant? How manj' stereocenters are present in FK-506? Determine the maximum number of stereoisomers possible. Identify and label the various functional groups present. Consider the two stereocenters in this structure labeled with asterisks (*). Determine the absolute configuration of each stereocenter. FK-506 has been shown to exhibit moderate solubility in various organic solvents. Is this immunosuppressant expected to be soluble in ethanol (CH3CH2OH)? Consider the carbon atom labeled “1." Describe the geometry and approximate bond angles about this carbon atom. Draw the alternative chair conformations of the cyclohexane ring at the lower right of FK-506 and label the more stable conformation. Are there any aromatic components present in FK-506? Patients taking FK-506 have reported several side effects from this medication, including headaches, nausea or diarrhea, and slight shaking. Would you expect the enantiomer of this drug to result in the same side effects?arrow_forward15-35 Following are structural formulas for three of the drugs most widely prescribed to treat depression. Label all stereocenters in each and state the number of stereoisomers possible for each. Fluoxetine (Prozac!arrow_forward15-8 What does the term “chiral” mean? Give an example of a chiral molecule.arrow_forward
- 15-11 In what way are constitutional isomers different from stereoisomers? In what way are they the same?arrow_forwardArachidonic acid is a naturally occurring C„o polyunsaturated fatty acid. Draw a line-angle formula for arachidonic acid showing the cis configuration about each double bond. CH2(CH2)4(CH=CHCH2)4CH2CH2COOH Arachidonic acidarrow_forward17-74 Glucose, C6H12O6, contains an aldehyde group but exists predominantly in the form of the cyclic hemiacetal shown here. We will discuss this cyclic form of glucose in Chapter 20. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. (a) Which carbon in glucose provides the —OH group and which provides the —CHO group? (b) Draw the alternative chair confirmations of D-glucose and state which of the two is the more stable.arrow_forward
- 15-9 What does the term “achiral” mean? Give an example of an achiral molecule.arrow_forward15-21 Answer true or false. For a molecule with two stereocenters, 22 = 4 stereoisomers are possible. For a molecule with three stereocenters, 32 = 9 stereoisomers are possible. Enantiomers, like gloves, occur in pairs. 2-Pentanol and 3-pentanol are both chiral and show enantiomerism. 1-Methylcyclohexanol is achiral and does not show enantiomerism. Diastereomers are stereoisomers that are not mirror images.arrow_forward15-43 Triamcinolone acetonide, the active ingredient in Azmacort Inhalation Aerosol, is a steroid used to treat bronchial asthma. Triamcinolone acetonide Label the eight stereocenters in this molecule. How many stereoisomers are possible for it? (Of these, the stereoisomer with the configura tion shown here is the active ingredient in Azmacort.)arrow_forward
- 15-30 (Chemical Connections 15A) What does it mean to say that a drug is chiral? If a drug is chiral, will it be optically active? That is, will it rotate the plane of polarized light?arrow_forward15-16 Which of the following compounds contain stereocenters? (a) 2-Chloropentane(b) 3-Chloropentane (c) 3-Chloro-l-butene(d) 1,2-Dichloropropanearrow_forward15-37 Consider a cyclohexane ring substituted with one hydroxyl group and one methyl group. Draw a structural formula for a compound of this composition that: Does not show cis-trans isomerism and has no stereocenters. Shows cis-trans isomerism but has no stereocenters. Shows cis-trans isomerism and has two stereocenters.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
