
Organic Chemistry - With Access (Custom)
4th Edition
ISBN: 9781259147760
Author: OHIO UNIV.
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15, Problem 15.22P
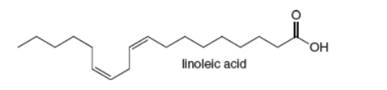
Which
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
2. Write down the specific form in which bromine (other than molecular bromine) could act as
(i) an electrophile : ………………………………..(ii) a free radical : …………………………………(iii) a nucleophile : ………………………………….
Draw the products expected from the addition of Bromine (Br2) to 1-heptyne.
Draw the structure of the major product of
2-pentyne + acidic KMnO4
Chapter 15 Solutions
Organic Chemistry - With Access (Custom)
Ch. 15 - Prob. 15.1PCh. 15 - Prob. 15.2PCh. 15 - Draw the product formed when a chlorine atom (Cl)...Ch. 15 - Prob. 15.4PCh. 15 - Prob. 15.5PCh. 15 - Problem 15.6 Using mechanism 15.1 as guide, write...Ch. 15 - Calculate m0 for the two propagation steps in the...Ch. 15 - Prob. 15.8PCh. 15 - Problem 15.8 Which bond in the each compound is...Ch. 15 - Prob. 15.10P
Ch. 15 - Prob. 15.11PCh. 15 - Synthesize each compound from (CH3)3CH. a....Ch. 15 - Prob. 15.13PCh. 15 - Prob. 15.14PCh. 15 - Prob. 15.15PCh. 15 - Prob. 15.16PCh. 15 - Prob. 15.17PCh. 15 - Prob. 15.18PCh. 15 - Draw all constitutional isomers formed when each...Ch. 15 - Draw the structure of the four allylic halides...Ch. 15 - Which compounds can be prepared in good yield by...Ch. 15 - Which CH bond is most readily cleaved in linolenic...Ch. 15 - Prob. 15.23PCh. 15 - Draw the products formed when each alkene is...Ch. 15 - Problem 15.24 When adds to under radical...Ch. 15 - Prob. 15.26PCh. 15 - Draw an energy diagram for the two propagation...Ch. 15 - Prob. 15.28PCh. 15 - Problem 15.27 Draw the steps of the mechanism that...Ch. 15 - Prob. 15.30PCh. 15 - Prob. 15.31PCh. 15 - Prob. 15.32PCh. 15 - Prob. 15.33PCh. 15 - Why is a benzylic CH bond labeled in red unusually...Ch. 15 - Prob. 15.35PCh. 15 - Prob. 15.36PCh. 15 - Prob. 15.37PCh. 15 - Prob. 15.38PCh. 15 - What alkane is needed to make each alkyl halide by...Ch. 15 - Which alkyl halides can be prepared in good yield...Ch. 15 - Prob. 15.41PCh. 15 - 15.40 Explain why radical bromination of p-xylene...Ch. 15 - a. What product(s) (excluding stereoisomers) are...Ch. 15 - Prob. 15.44PCh. 15 - Prob. 15.45PCh. 15 - Prob. 15.46PCh. 15 - 15.44 Draw all constitutional isomers formed when...Ch. 15 - Draw the organic products formed in each reaction....Ch. 15 - Prob. 15.49PCh. 15 - 15.47 Treatment of a hydrocarbon A (molecular...Ch. 15 - Prob. 15.51PCh. 15 - Prob. 15.52PCh. 15 - Prob. 15.53PCh. 15 - Prob. 15.54PCh. 15 - 15.53 Consider the following bromination: .
a....Ch. 15 - 15.54 Draw a stepwise mechanism for the following...Ch. 15 - Prob. 15.57PCh. 15 - An alternative mechanism for the propagation steps...Ch. 15 - Prob. 15.59PCh. 15 - Prob. 15.60PCh. 15 - Devise a synthesis of each compound from...Ch. 15 - Devise a synthesis of each target compound from...Ch. 15 - Devisea synthesis of each target compound from the...Ch. 15 - Devise a synthesis of each compound using CH3CH3...Ch. 15 - Prob. 15.65PCh. 15 - 15.63 As described in Section 9.16, the...Ch. 15 - 15.64 Ethers are oxidized with to form...Ch. 15 - Prob. 15.68PCh. 15 - Prob. 15.69PCh. 15 - 15.67 In cells, vitamin C exists largely as its...Ch. 15 - What monomer is needed to form each...Ch. 15 - Prob. 15.72PCh. 15 - Prob. 15.73PCh. 15 - 15.71 Draw a stepwise mechanism for the following...Ch. 15 - 15.72 As we will learn in Chapter 30, styrene...Ch. 15 - Prob. 15.76PCh. 15 - 15.74 A and B, isomers of molecular formula , are...Ch. 15 - Prob. 15.78PCh. 15 - Radical chlorination of CH3CH3 forms two minor...Ch. 15 - 15.76 Draw a stepwise mechanism for the...Ch. 15 - Prob. 15.81PCh. 15 - Prob. 15.82PCh. 15 - Prob. 15.83P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The strength of the HO bond in vitamin E is weaker than the CH from which the hydrogen is abstracted in the fatty acid primarily because, 1. OH bonds are always weaker than CH bonds. 2. The radical created from hydrogen abstraction from OH is stabilized by resonance delocalization and many of the contributing structures have secondary carbon radical character. 3. The sp2 hybridization of the phenyl ring changes the hybridization of the oxygen in the OH bond. 4. The radical created from hydrogen abstraction from OH is stabilized by resonance delocalization and many of the contributing structures have tertiary carbon radical character.arrow_forwardDraw the structure of the organic product expected when BrCH2CH(CH3)CH2OH is treated with base.arrow_forwardCan 4-methylcyclohexanoic acid be reduced by NABH4?arrow_forward
- In cells, vitamin C exists largely as its conjugate base X. X is an antioxidant because radicals formed in oxidation processes abstract the labeled H atom, forming a new radical that halts oxidation. Draw the structure of the radical formed by H abstraction, and explain why this H atom is most easily removed.arrow_forwardDraw resonance structures for the carbocation formed after loss of a leaving group from phytyl diphosphate.arrow_forwardWhich C–H bond is most readily cleaved in linoleic acid? Draw all possible resonance structures for the resulting radical. Draw all the hydroperoxides formed by reaction of this resonance-stabilized radical with O2.arrow_forward
- Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.arrow_forwardDraw the structure of the products that will be formed when aniline reacts with nitrous acid at 5 oC,followed by:arrow_forwardThe conducted experiment: In a 25 mL rbf add 5 mL of saturated sodium bicarbonate (NahCO3) , 0.5 mL trimethylphosphonoacetate and 0.386g of your 4-chlorobenzaldehyde. Write the mechanism of the reaction you performed. What is the structure of the water - soluble dialkylphosphate salt formed.--------------------------------------------- An example is provided that used trimethylphosphonate, sodium methoxide, methanol and 3-chlorobenzaldehdye. IT IS ONLY AN EXAMPLE.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY