
Concept explainers
Interpretation:
Four isomers consistent with the formula
Concept introduction:
The IHD accounts for the number of rings or multiple bonds the structure could possess.
Number of conjugated C=C double bond are predicted using following table.
| Conjugated |
UV |
Change in |
| - | ||
Answer to Problem 15.32P
Four isomers consistent with the formula
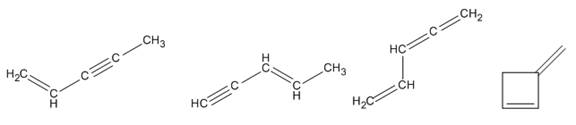
Explanation of Solution
The given molecular formula is
From the table
| Conjugated |
UV |
Change in |
| - | ||
The two conjugated
Therefore four isomers consistent with the formula
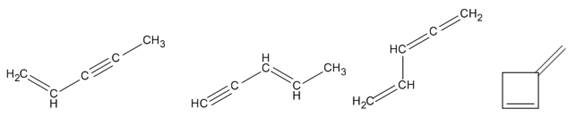
Isomers consistent with the formula
Want to see more full solutions like this?
Chapter 15 Solutions
ORGANIC CHEMISTRY:PRINCIPLES...(CL)
- Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.)E,Z isomerism but not chiralityarrow_forwardI have the following data from an NMR scan. Five peaks; a doublet at about 0.8ppm (6H) a small single septet at about 1.5 ppm (1H), a triplet at 1.6 ppm (2H(, a singlet at about 2.1 ppm (3H), a triplet at about 4.4 ppm (2H). The molecular formula is C7H14O2. I calculated a degree of unsaturation of 1. I have tentatively identified the compound as methyl-4-methylbutanoate. I would appreciate any comments you care to make. Thank you.arrow_forwardDraw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.Chirality but not E,Z isomerismarrow_forward
- Draw and name each of the following: a. An ether with molecular formula C3H8O b. An alkene with the formula C5H10 that has stereoisomers. c. A tertiary (30) alcohol with molecular formula C5H11OH that is chiral. d. A primary (10) aliphatic amine with molecular formula C3H9 e. A thiol with molecular formula C6H14arrow_forwardDraw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.Both chirality and E,Z isomerismarrow_forward5. Compare the absorption wavelength of two compounds:1,3-cyclohexadiene and 1,4-cyclohexadiene. Which of the two compounds will show UV absorption at a lower wavelength? What is the possible explanation for that? Include the reference usedarrow_forward
- which of the following cannot be distinguished by an infrared spectrum a) resonant hybrids b) tautomers (keto-enol) c) enantiomers d) cis-trans isomersarrow_forwardIn addition to more highly fluorinated products, fluorination of 2-methylbutane yields a mixture of compounds with the formula C5H10F2. Draw the structures of all the isomers with the formula C5H10F2 that would be produced and label with a star all the chiral centers present in their structures.arrow_forwardWill you help me find all the ethers with the formula: C5H12O ? I also need to provide the common and systemic names.arrow_forward
- Identify the structures of A and B, isomers of molecular formula C10H12O2, from their IR data and 1H NMR spectra.a. IR absorption for A at 1718 cm−1b. IR absorption for B at 1740 cm−1arrow_forwardAn IR spectrum, a 13C NMR spectrum, and a 1H NMR spectrum were obtained for an unknown structure with a molecular formula of C8H18O. Draw the structure of this compound.arrow_forwardCalculating the Number of Degrees of Unsaturation in Compounds with O, X, or N Calculate the number of degrees of unsaturation for each molecular formula: (a) C5H8O; (b) C6H11Cl; (c) C8H9N. Propose one possible structure for each compound.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
