
Concept explainers
(a)
Interpretation:
A concept map is to be drawn and the grams of
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 19E
The concept map is shown below.
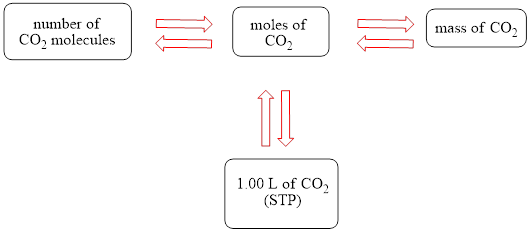
The grams of
Explanation of Solution
When

Figure 1
The volume occupied by
Therefore, the number of moles which occupy
The molar mass of
Therefore, the mass of
The formula to calculate the mass of
Substitute the mass of
Therefore, the grams of
The grams of
(b)
Interpretation:
A concept map is to be drawn and the molecules of
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 19E
The concept map is shown below.

The molecules of
Explanation of Solution
When

Figure 1
The volume occupied by
Therefore, the number of moles which occupy
The molecules present in
The formula to calculate the molecules occupied by
Substitute the molecules in
Therefore, the molecules of
The molecules of
(c)
Interpretation:
A concept map is to be drawn and the molar concentration of the carbonic acid solution when
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 19E
The concept map is shown below.

The molar concentration of the carbonic acid solution is
Explanation of Solution
When

Figure 1
The volume occupied by
Therefore, the number of moles which occupy
The number of moles in
The relation between
The probable unit factors are given below.
The unit factor to determine
Therefore, the volume in
The formula to determine molarity is shown below.
Where
•
•
•
Substitute the value of number of moles and volume in equation (1).
The relation between
The unit factors are given below.
The unit factor to determine
Therefore,
Therefore, the molar concentration of
The molar concentration of carbonic acid solution is
Want to see more full solutions like this?
Chapter 15 Solutions
EP INTRODUCTORY CHEM.-MOD.MASTERINGCHEM
- Mixtures of gases are always true solutions. True or false? Explain why.arrow_forwardCalculate the number of grams of solute in each of the following solutions. (a) 4.84 L of a 1.46 M HCl solution grams of HCl (b) 31.7 mL of a 11.3 M HNO3 solution grams of HNO3 (c) 57.6 mL of a 10.3 M AgNO3 solution grams of AgNO3 (d) 1.54 L of a 3.4×10-3 M Na2SO4 solution grams of Na2SO4arrow_forwardWhat is the mass of a 5.6 liter (STP) sample of CO2 gas?arrow_forward
- 1. How would each of the following procedural errors affect the value obtained for the molar volume of hydrogen gas? Explain your reasoning. (a) Some bubbles of hydrogen gas remained clinging to the sides of the tube. (b) Some magnesium metal was left un-reacted at the end of the experiment. (c) 7 mL of the HCl were used instead of 5 mL.arrow_forwardCalculate the grams of solute in each of the following solutions: (a) 3.4 L of 0.87 M K2CrO4 (b) 70.7 mL of 0.037 M HC2H3O2 (c) 240 . mL of 16 M HNO3arrow_forwardDetermine the molarity for each of the following solutions: (d) 10.5 kg of Na2SO4·10H2O in 18.60 L of solutionarrow_forward
- 2. Hydrochloric acid (HCI) can dissolve solid iron according to the following reaction: Fe (s) + 2 HCI (aq) → FeCl2 (aq) + H2 (g) a) How many moles of HCl are needed to completely dissolve a 210 g iron bar? b) What volume of H2 gas will be produced by the complete reaction of the iron bar? Assume STP.arrow_forwardSuppose that 0.3744 L of 1.056 mol / L of barium chloride is added to 0.5744 L of 1.356 mol / L of sodium sulfate (excess). Barium chloride reacts with sodium sulfate as per the equation above. To calculate the number of moles of barium chloride which are present initally before the reaction, you should use the following formula: n = c x v The number of mole value is:arrow_forwardA reagent bottle contains 6.00 M NaOH solution. How many gram of NaOH must be dissolved to prepare 473 mL of such solution? (A) 71.0 g (B) 114 g (C) 507 g (D) 3150 garrow_forward
- How many grams of solute are present in 855 mL of 0.880 M KBr?arrow_forwardHow many milliters of a 3.0 M H3PO4 solution are required to react with excess zinc to form 145g of H2 gas?arrow_forwardWhat volume of 0.350 M HNO3 Can be made from 150mL of 6.6 M HNO3? L (Give answer to correct sig figs in the form of units of L)arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

