
EBK ORGANIC CHEMISTRY
10th Edition
ISBN: 9781259636875
Author: Carey
Publisher: MCGRAW HILL BOOK COMPANY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 34P
Like other hydroborations, the reaction of
and its regioselectivity is opposite to Markovnikov's rule.
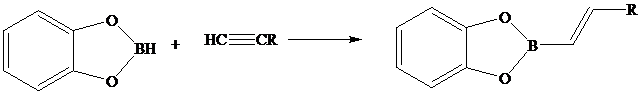
Use this fact to outline a synthesis of the compound shown from
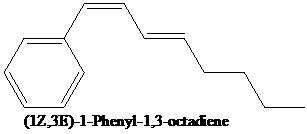
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
name the two diene products that could be obtained in the double-dehydration reaction of hexane-2,4-diol, and identify the major product.
Give the structures of the substitution products expected when 1-bromohexane reacts with(a) NaOCH2CH
Write the structure of the major organic product isolated from the reaction of 3-hexyne with (a) Hydrogen (2 mol), platinum (b) Hydrogen (1 mol), Lindlar palladium (c) Hydrogen chloride (1 mol) (d) Hydrogen chloride (2 mol) (e) Chlorine (1 mol) (f) Chlorine (2 mol) (g) Aqueous sulfuric acid, mercury(II) sulfate (h) Ozone followed by hydrolysis
Chapter 15 Solutions
EBK ORGANIC CHEMISTRY
Ch. 15.1 - Each of the following organometallic reagents will...Ch. 15.3 - Write equations showing how you could prepare...Ch. 15.4 - Lithium diisopropylamide is often used as a strong...Ch. 15.5 - Write the structure of the organic product of each...Ch. 15.7 - Prob. 5PCh. 15.8 - Prob. 6PCh. 15.9 - Prob. 7PCh. 15.9 - Like nickel, iron reacts with carbon monoxide to...Ch. 15.9 - Prob. 9PCh. 15.9 - What is the oxidation state of manganese in the...
Ch. 15.9 - Prob. 11PCh. 15.10 - Prob. 12PCh. 15.10 - Prob. 13PCh. 15.11 - Give the structure including stereochemistry of...Ch. 15.11 - Prob. 15PCh. 15.12 - Homogeneous catalytic hydrogenation of the...Ch. 15.12 - Prob. 17PCh. 15.13 - What alkenes are formed from 2-pentene by olefin...Ch. 15.13 - The product of the following reaction was isolated...Ch. 15 - Suggest appropriate methods for preparing each of...Ch. 15 - Prob. 21PCh. 15 - Predict the principal organic product of each of...Ch. 15 - Prob. 23PCh. 15 - Predict the principal organic product of each of...Ch. 15 - Prob. 25PCh. 15 - A different stereoisomer of...Ch. 15 - Prob. 27PCh. 15 - Using phenyllithium and any necessary organic or...Ch. 15 - Prob. 29PCh. 15 - A number of drugs are prepared by reactions in...Ch. 15 - The following conversion was carried out in two...Ch. 15 - Outline syntheses of (a)...Ch. 15 - (S)-(+)-Ibuprofen can be prepared by...Ch. 15 - Like other hydroborations, the reaction of alkynes...Ch. 15 - The sex attractant of the female silkworm has been...Ch. 15 - Prob. 36PCh. 15 - (a) Exaltolide, a musk substance, has been...Ch. 15 - Prob. 38PCh. 15 - Prob. 39PCh. 15 - Cyclobutadiene and...Ch. 15 - Cyclobutadiene and (Cyclobutadiene)tricarbonyliron...Ch. 15 - Cyclobutadiene and...Ch. 15 - Cyclobutadiene and...Ch. 15 - Cyclobutadiene and...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- When 5-bromo-1-pentanol is treated with sodium hydride in diethyl ether, the product is analyzed to be C5H10O. Propose a likely structure for this product, suggesting a reasonable mechanistic pathway for its formationarrow_forwardWhat products would be obtained from the reaction of 1,3,5-hexatriene with one equivalent of HBr? Disregard stereoisomers.arrow_forwardWhat product would you expect to obtain from addition of Cl2to 1, 2-dimethylcyclohexene? Show the stereochemistry of the productarrow_forward
- Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.)2,4,6-Trinitrotoluene (TNT)arrow_forwardThe carbocation electrophile in a Friedel-Crafts reaction can be generated in ways other than by reaction of an alkyl chloride with AlCl3. For example, reaction of benzene with 2-methylpropene in the presence of H3PO4 yields tert-butylbenzene. Draw a structure for the electrophile.arrow_forwardBenzene, C6H6 undergoes substitution reaction with concentrated nitricacid, HNO3 to produce compound L. The reaction of compound L withbromine, Br2 in the presence of iron tribromide, FeBr3 produced compoundM. Benzene also undergoes Fridel-crafts alkylation reaction withchloroethane, CH3CH2Cl using catalyst N to produce compound P (i) Draw the structural formula of L, M and P (ii) State catalyst N. (iii) Show the formation of electrophile that will be reacted with benzene for theformation of compound P.arrow_forward
- Suppose you were told that each reaction is a substitution reaction, but you were not told the mechanism. Describe how you could conclude from the structure of the haloalkane or cycloalkene, the nucleophile, and the solvent that each reaction is an SN1 reaction.arrow_forwarda) Give the appropriate base for the above reaction and draw the resonance structures of the enolate ions derived from compound A. Then write the mechanism for the formation of the Dieckmann cyclized product B. b) If compound B above is reacted with NaBH 4 , draw the structure of the reduced product. Give a reason for your choice of product. c) Being a β-ketoester, B could undergo a 3-step synthesis involving alkylation, hydrolysis and decarboxylation reactions to yield the cyclopentanone, C. Write the outline synthesis for each step which include the appropriate regents and correct intermediate.arrow_forward(10pts) Compound A, C10H16, was found to be optically active. On catalytic reduction over a palladium catalyst, 2 equivalents of hydrogen were absorbed, yielding compound B, CioH2o. On ozonolysis of A, two fragments were obtained. One fragment was identified as acetic acid (CHCOOH). The other fragment, compound C, was an optically active carboxylic acid, C8H14O2. Write reactions, and draw the correct structures for A-C, explain your answer in detail.arrow_forward
- 1. Compound A, C9H12, absorbs 3 equivalents of H2 on catalytic hydrogenation over palladium catalyst to give B, C9H18. On the treatment with acidic KMnO4, compound A gives among other things, a ketone that was identified as cyclohexanone. On reaction with NaNH2 in NH3, followed by addition of iodomethane, compound A gives a new hydrocarbon C, C10H14. What are the structures of A , B and C?arrow_forwardBased on what you know about the relative stabilities of alkyl cations and benzyliccations, predict the product of addition of HBr to 1-phenylpropenearrow_forwardDescribe a sequence of reactions by which hept-1-yne can be straightforwardly prepared from hept-1-ene.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License