
Concept explainers
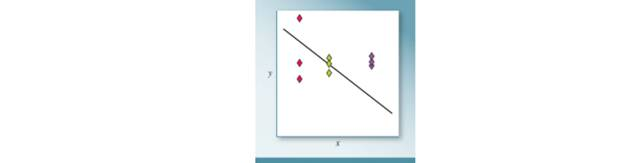
The true dependence of y on x is represented by the line. Three students measured y as a function of x and plotted their data on the graph. Which set of data has the best accuracy and which has the best precision, respectively?
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Chemistry - With Access (Looseleaf) (Custom)
- What is the percentage uncertainty with the following measurement 0.100 ± 0.005 g? Provide your answer to one decimal point.arrow_forwardThe accepted value of the melting point of pure aspirin is 135 °C. Trying to verify that value, you obtain 134 °C, 136 °C, 133 °C, and 138 °C in four separate trials. Your partner finds 138 °C, 137 °C, 138 °C, and 138 °C. (a) Calculate the average value and percent error for your data and your partner’s data. (b) Which of you is more precise? More accurate?arrow_forwardA Chemistry 20 student uses a thermometer and a hot plate and measures the boiling point of ethyl alcohol to be 74.1 ºC. Then, she looks in a reference book and finds that the actual boiling point of ethyl alcohol is 78.4 ºC. What is her percent error?Record only your final answer with the correct number of significant digits and the proper units.arrow_forward
- A Chemistry 20 student uses a thermometer and a hot plate and measures the boiling point of ethyl alcohol to be 71.1 ºC. Then, she looks in a reference book and finds that the actual boiling point of ethyl alcohol is 78.4 ºC. What is her percent error?Do not show your work in the space provided.Record only your final answer with the correct number of significant digits and the proper units.arrow_forwardConsider the addition of “1 5.4” to “28.” What would a mathematician say the answer is? What would a scientist say? justify the scientist’s answer, not merely citing the rule, but explaining it.arrow_forwardIf you repeat the same measurement many times, will you always obtain exactly the same result? Why or why not? What factors influence the repeatability of a measurement?arrow_forward
- In lab you report a measured volume of 128.7 mL of water. Using significant figures as a measure of the error, what range of answers does your reported volume imply? Explain.arrow_forward1.17 Complete the following statement: Data that have a large systematic error can still he (a) accurate, (b) precise, or (c) neither.arrow_forwardEach of the following coins is photographed to scale. Measure each using a metric ruler (in cm) and record the diameter to the precision reflected in the ruler. Make a simple graph of the value of the coin (x-axis) and the diameter of the coin (y-axis). Is there any correlation between these two quantities? Which coin does not fit the general trend?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning  Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





