
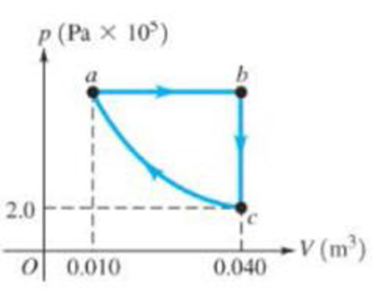
Figure 15.40
Problem 76.
The graph in Figure 15.40 shows a pV diagram for 3.25 moles of ideal helium (He) gas. Part ca of this process is isothermal. (a) Find the pressure of the He at point a. (b) Find the temperature of the He at points a, b, and c. (c) How much heat entered or left the He during segments ab, bc. and ca? In each segment, did the heat enter or leave? (d) By how much did the internal energy of the He change from a to b, from b to c, and from c to a? Indicate whether this energy increased or decreased.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
College Physics Plus Mastering Physics with eText - Access Card Package (10th Edition)
Additional Science Textbook Solutions
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Essential University Physics (3rd Edition)
University Physics (14th Edition)
Cosmic Perspective Fundamentals
Sears And Zemansky's University Physics With Modern Physics
- If you have 3 moles of a monoatomic ideal gas, how much heat is required to raise the temperature of this gas from 263.3K to 276.1K if the volume of the gas remains constant during the heating? Note: It is understood that your answer is in units of Joules, however do not explicitly include units in your answer. Enter only a number. If you do enter a unit ("J" in this case), you answer will be counted wrong.arrow_forwardA monatomic ideal gas undergoes an isothermal expansion at 300 K, as the volume increased from 0.020 to [2*v1] m³.The final pressure is 120.61kPa. The ideal gas constant is R = 8.314 J/mol · K. %3D What is the heat transfer Q to the gas in kilojoules? Please enter your numerical answer with two decimal places. (for example for 2090 J, enter 2.09 )arrow_forwardThe fpicture shows a pV diagram for an ideal gas in which its pressure tripled from a to b when 804 J of heat was put into the gas. a) How much work was done on or by the gas between aa and bb? Express your answer in joules. b) Fill in the sentence, do they increase, decrease, or stay the same. " The temperature of this gas ______ between a and b. When pressure increases, the temperature _____ provided that V and n are _______." c) By how much did the internal energy of the gas change between a and b? Express your answer in joules. Did it increase or decrease? d) What is the temperature of the gas at point b in terms of its temperature at a, Taarrow_forward
- A gas expands from I to F in the figure below. The energy added to the gas by heat is 276 J when the gas goes from I to F along the diagonal path. A pressure-volume graph consists of points and line segments plotted on a coordinate plane, where the horizontal axis is V (liters)and the vertical axis is P (atm). Three points are plotted: point I at (2, 4) point A at (4, 4) point F at (4, 1) Line segments connect the three points to form a triangle. Arrows along the line segments point from I to A, from A to F, and from I to F. (a) What is the change in internal energy of the gas? J (b) How much energy must be added to the gas by heat along the indirect path IAF?arrow_forwardProblem 2: The enthalpy of a system is given by the equation H = U + PV where U is the internal energy, P = pressure, and V = volume. In addition, the internal energy, U = Q + W where Q is the heat and W is the work. Suppose we want to find the rate of change in the enthalpy at constant pressure of 1.25 atm, what is the value when heat is absorbed by the system at a rate of 45 J/s and work is done by the system at a rate of 100 J/s when the change of volume is rated at 35 x 105 m/s? 1. What is the change in heat with respect to time? 2. What is the change in internal energy of the system with respect to time? 3. What is the change in enthalpy of the system with respect to time?arrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 422 J when the gas goes from I to F along the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V (liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B (2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I (2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F (4,1). (a) What is the change in internal energy of the gas? J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in internal energy? Jarrow_forward
- The temperature at state A is 20.0°C, that is 293 K. During the last test, you have found the temperature at state D is 73.0 K and n = 164 moles for this monatomic ideal gas. What is the change in thermal energy for process A to D, in MJ (MegaJoules)? Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. P (atm) 4 3 ID B +V (m) 4 5arrow_forwardIn a certain process, the energy of the system decreases by 250 kJ. The process involves 480 kJ of work done on the system. Find the amount of heat Q transferred in this process. Express your answer numerically in kilojoules. Make your answer positive if the heat is transferred into the system; make it negative if the heat is transferred into the surroundings.arrow_forwardThe heat engine shown in the figure uses 2.0 mol of a monatomic gas as the working substance. (Figure 1) Figure p (kPa) 600 400 200 0 0 0.025 0.050 V (m³) 1 of 1 Determine T₁, T2, and T3. Enter your answers numerically separated by commas. Express your answer using two significant figures. T₁, T2, T3 Submit ✓ Correct Part B 600,1800,1200 K Previous Answers Determine AEth, Ws, and Q for 1-2. Enter your answers numerically separated by commas. Express your answer using two significant figures. 15. ΑΣΦ AEth, Ws, Q = 30, 12.5,42.5 Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining ? Jarrow_forward
- A container is filled with an ideal diatomic gas to a pressure and volume of P₁ and V₁, respectively. The gas is then warmed in a two-step process that increases the pressure by a factor of four and the volume by a factor of five. Determine the amount of energy transferred to the gas by heat if the first step is carried out at constant volume and the second step at constant pressure. (Use any variable or symbol stated above as necessary.) Q =arrow_forwardm = 0.593 kg of methanol in atmospheric pressure is heated with an electric heater from T₁ = 30.2 °C to its boiling point. After the heating my methanol is vaporized. Heating and vaporizing takes t = 450.41 s with n=0.59 heater's efficiency. What is the volume of methanol? V = What is the energy required for heating? E₁ = 1 kJ m³ Your last answer was interpreted as follows: 1 What is the energy required for vaporizing? Ev = 1 kJ Your last answer was interpreted as follows: 1 What is the power of the heater? P = 1 с Your last answer was interpreted as follows: 1 Your last answer was interpreted as follows: 1 What is the electr. energy needed for heating and vaporizing? Eε = 1 kJ Your last answer was interpreted as follows: 1 h Do not leave any fields blank. If you don't know the answer, insert 1 for example. Insert the answer with 3 significant digits without rounding the answer. Constant P Tb Value Unit 2.53 1165 786.3 64.7 °C umges Quantity heat capacity kg K kJ kW kg kg LAN Symbol h…arrow_forwardThe heat engine shown in the figure uses 2.0 mol of a monatomic gas as the working substance. (Figure 1) Figure p (kPa) 600 400 200 0 0 0.025 0.050 < 1 of 1 V (m³) Part D Determine AEth, Ws, and Q for 3→1. Enter your answers numerically separated by commas. Express your answer using two significant figures. VE ΑΣΦ AEth, Ws, Q = Submit Part E Request Answer What is the engine's thermal efficiency? Express your answer using two significant figures. ? Jarrow_forward
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
