
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
Consider the reaction
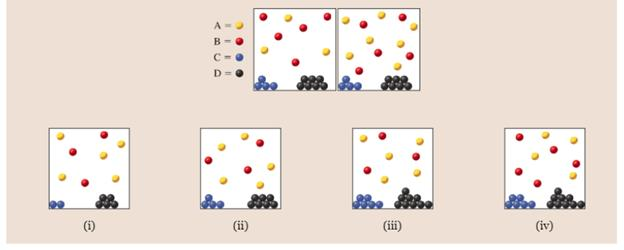
where 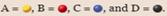 . The right side of the first diagram represents the system immediately after more A has been added to stress the equilibrium. Which of the following diagrams [(i)-(iv)] could represent the system after equilibrium has been reestablished? Select all that apply.
. The right side of the first diagram represents the system immediately after more A has been added to stress the equilibrium. Which of the following diagrams [(i)-(iv)] could represent the system after equilibrium has been reestablished? Select all that apply.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
ALEKS 360-ACCESS (1 SEMESTER)
- Carbon monoxide and hydrogen react according to the following equation: CO (g) + H2 (g) CH4 (g) + H2O (g) When 1.00 mol of CO and 3.00 moles of H2 are placed in a 10.0 L container at 927 ° C (1200 K) and allowed to reach equilibrium, the mixture is found to have 0.363 moles of H2O What is the molar composition (that is, how many moles) of H2 are present in the equilibrium mixture? Answer:arrow_forwardReaction of nitrogen and hydrogen in the presence of a catalyst produces ammonia: N2 + 3H2 2NH3 , rHo = -92 kJ mol-1 . What would increase equilibrium concentration of the product in the mixture? A) Increase pressure, keep temperature constant B)Increase temperature, keep pressure constant C) Decrease pressure, keep temperature constant D) Information is insufficient for answerarrow_forwardSolve all part please. Consider the equilibrium reaction N2O4 (g) ⇌ 2 NO2 (g)a. Using appendix C, find H and S for this reaction (you will need this for b,c). delta s= 176.6J delta H=5802kJb. Knowing that K is temperature dependent and that G = -RTlnK, find the equilibrium constant (K) and T whenthe above reaction is at equilibrium with PNO2 = PN2O4 = 1.0 atm.c. Knowing that K is temperature dependent and that G = -RTlnK, find the equilibrium constant (K) and T whenthe above reaction is at equilibrium with PT = 10.0 atm and PNO2 = 2 PN2O4.arrow_forward
- What is ΔG of the following reaction at 32°C and the reaction quotient is 2.45 (ΔG°=-938.7 kJ/mol). -936.43 kJ/mol 1333.6 kJ/mol -876.65 kJ/mol 954.53 kJ/mol Not enough informationarrow_forwardConsider the following reaction: CO (g) + H2O (g) <===> CO2 (g) + H2 (g) a. Write the expression for the equilibrium constant (K) of the reaction b. Calculate the value for K using the following concentrations of each substance at equilibrium: [CO) = 0.55 M [H2O) = 0.44 M [CO2) = 0.25 M [H2) = 0.10 M c. Based on the value you obtained for K, are the products or reactants lower in energy? d. Is the value for ΔH negative or positive? please show workarrow_forwardSuppose you are given the reaction A(g) + B(g) C(g) + D(g). You have the gases A, B, C, and D at equilibrium. Upon adding gas B, the value of K: Select one: a. increases, because by adding A more products are made, increasing the product to reactant ratio b. does not change, as long as the temperature is constant c. depends on whether the reaction is endothermic or exothermic d. does not change, because A does not figure into the product to reactant ratio e. decreases, because A is a reactant so the product to reactant ratio decreasesarrow_forward
- For the equilibrium A + B ⇌ C + D K was measured to be 1.51 × 10⁻² at 25°C and 15.0 at 600°C. What is ∆S° for the reaction? Assume ∆H° and ∆S° are temperature independent. Answer in J/mol x Karrow_forwardFor the reaction: 2 A (g) + B (s) ⇌ 2 C (s) + D (g) At 298 K in a 10.0 L vessel, the equilibrium values are as follows: 0.721 atm of A, 4.18 mol of B, 6.25 mol of C, and 2.91 atm of D. What is the value of the equilibrium constant? Answer _____arrow_forwardChemistry Find ΔHfus for water Mass of cup and warm water: 45.67 gFinal mass of cup, water, and melted ice: 69.76 gMass of empty cup: 2.21 gTemperature of warm water: 63 °CTemperature of ice: 0 °CTemperature at equilibrium: 45.2 °Carrow_forward
- The reaction shown below has an equilibrium constant value of Kp=5.50 at a certain temperature.CO (g) + Cl₂ (g) ⇄ COCl₂ (g) Kp = 5.50A container is pressurized with CO and Cl₂ until the pressure of CO is 0.450 atm and the pressure of Cl₂ is 0.200 atm. Calculate the pressure of COCl₂ in the container when the system reaches equilibrium.Answer: atm COCl₂arrow_forward4.4 g of CO2(g) are introduced into a 1 dm3 flask containing excess carbon at 1000oC, so that the equilibrium CO2(g) + C(s) 2 CO(g) is reached. The gas density at equilibrium corresponds to an average molecular weight of 36. Calculate the equilibrium total pressure and the value of Kp. Answer: [1408 kPa; 7.04]arrow_forwardA reaction vessel is charged with 0.50 atm of A and 0.270 atm of B. Once the reaction reaches equilibrium, what is the equilibrium partial pressure of B? Kp for this reaction is 67.2 A (g) ⇌ 2 B (g) Answer : _____ atmarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

