
Aleks 360 2 Semester Access Card For Chemistry
4th Edition
ISBN: 9781259626685
Author: Julia Burdge
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15.13, Problem 1PPC
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
Consider the reaction
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
issue 6Choose the equilibrium in which the products are favored by a drop in pressure and the reactants favored by a drop in temperature.a) N2(g) + 3 H2(g) ⇔ 2 NH3(g) + 92.3 kJb) H2(g) + I2(g) + 51.8 kJ ⇔ 2 HI(g)c) PCl3(g) + Cl2(g) ⇔ PCl5(g) ΔH = -84.2 kJd) 2 NO2(g) ⇔ 2 NO(g) + O2(g) ΔH = +54 kJ
A chemical equilibrium A ⇄ 2B has a forward rate constant, kf = 12 M –1 s–1, and a reverse rate constant, kr = 18 s–1. If the system has a concentration of [A] = 0.15 M at equilibrium, what is the concentration of B at equilibrium? (no scientific notation)
Consider the hypothetical reaction, which has an equilibrium constant (Kp) of 16.1 when the reaction occurs at 167°C.
2 W (g) + X (g) <---> 3 Y (g)
If the equilibrium partial pressure of W is 0.787 atm and that of Y is 1.65 atm, what will the equilibrium partial pressure (in atm) of X be?
(answer in 3 significant figures)
Chapter 15 Solutions
Aleks 360 2 Semester Access Card For Chemistry
Ch. 15.1 - Practice Problem ATTEMPT
In an analysis of the...Ch. 15.1 - Prob. 1PPBCh. 15.1 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.2 - Practice ProblemATTEMPT Write the reaction...Ch. 15.2 - Practice Problem BUILD
Write the equation for the...Ch. 15.2 - Practice ProblemCONCEPTUALIZE In principle, in the...Ch. 15.2 - Select the correct equilibrium expression for the...Ch. 15.2 - Prob. 2CPCh. 15.3 - Practice Problem ATTEMPT Write equilibrium...Ch. 15.3 - Practice Problem BUILD
Which of the following...
Ch. 15.3 - Prob. 1PPCCh. 15.3 - Prob. 1CPCh. 15.3 - Prob. 2CPCh. 15.3 - Given the following information: HF ( a q ) ⇄ H +...Ch. 15.3 - Prob. 4CPCh. 15.4 - Practice ProblemATTEMPT The following reactions...Ch. 15.4 - Practice Problem BUILD
The equation represents a...Ch. 15.4 - Practice ProblemCONCEPTUALIZE Consider a chemical...Ch. 15.4 - Use the following information to answer questions...Ch. 15.4 - Prob. 2CPCh. 15.4 - 15.4.3 If for the reaction at a certain...Ch. 15.4 - If K c = 3 for the reaction X + 2Y ⇄ Z at a...Ch. 15.5 - Practice ProblemATTEMPT Write K? expressions for (...Ch. 15.5 - Prob. 1PPBCh. 15.5 - Prob. 1PPCCh. 15.5 - Prob. 1CPCh. 15.5 - Prob. 2CPCh. 15.5 - Prob. 3CPCh. 15.5 - Prob. 4CPCh. 15.5 - Prob. 5CPCh. 15.5 - Prob. 6CPCh. 15.6 - Practice Problem ATTEMPT
For the reaction:
....Ch. 15.6 - Practice ProblemBUILD K p = 2.79 × 10 − 5 for the...Ch. 15.6 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.7 - Prob. 1PPACh. 15.7 - Prob. 1PPBCh. 15.7 - Prob. 1PPCCh. 15.8 - Practice ProblemATTEMPT Calculate the equilibrium...Ch. 15.8 - Practice ProblemBUILD Determine the initial...Ch. 15.8 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.9 - Prob. 1PPACh. 15.9 - Prob. 1PPBCh. 15.9 - Prob. 1PPCCh. 15.10 - Practice ProblemATTEMPT Aqueous hydrocyanic acid...Ch. 15.10 - Practice Problem BUILD Consider a weak acid, HA,...Ch. 15.10 - Practice ProblemCONCEPTUALIZE Each of the...Ch. 15.11 - Practice Problem ATTEMPT Determine the equilibrium...Ch. 15.11 - Prob. 1PPBCh. 15.11 - Prob. 1PPCCh. 15.12 - Practice ProblemATTEMPT For each change indicated,...Ch. 15.12 - Prob. 1PPBCh. 15.12 - Practice ProblemCONCEPTUALIZE Consider the...Ch. 15.13 - Practice Problem ATTEMPT
For each reaction,...Ch. 15.13 - Practice Problem BUILD
For the following...Ch. 15.13 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.14 - Practice Problem ATTEMPT
The reaction of carbon...Ch. 15.14 - Practice Problem BUILD
Consider the hypothetical...Ch. 15.14 - Practice Problem CONCEPTUALIZE
The decomposition...Ch. 15 - The K a for hydrocyanic acid ( HCN ) is 4 .9 × 10...Ch. 15 - 15.2
Determine the concentrations of in a...Ch. 15 - 15.3
Determine the for a weak acid if a 0.10-M...Ch. 15 - Prob. 4KSPCh. 15 - Define equilibrium. Give two examples of a dynamic...Ch. 15 - 15.2 Which of the following statements is correct...Ch. 15 - 15.3 Consider the reversible reaction Explain how...Ch. 15 - What is the law of mass action?Ch. 15 - Briefly describe the importance of equilibrium in...Ch. 15 - Define reaction quotient. How does it differ from...Ch. 15 - Write reaction quotients for the following...Ch. 15 - Write the equation for the reaction that...Ch. 15 - Consider the reaction: 2NO ( g ) + 2H 2 ( g ) ⇄ N...Ch. 15 - The equilibrium constant for the reaction: 2SO 2 (...Ch. 15 - Consider the following equilibrium process at...Ch. 15 - The equilibrium constant for the reaction: 2 H 2 (...Ch. 15 - The first diagram represents a system at...Ch. 15 - These two diagrams represent systems at...Ch. 15 - Define homogeneous equilibrium and heterogeneous...Ch. 15 - What do the symbols K c and K p represent?Ch. 15 - Write the expressions for the equilibrium...Ch. 15 - Write equilibrium constant expressions for K c ,...Ch. 15 - Write the equilibrium constant expressions for K c...Ch. 15 - 15.20 Write the equation relating to , and define...Ch. 15 - 15.21 The equilibrium constant () for the...Ch. 15 - What is K p at 1273°C for the reaction 2CO ( g ) +...Ch. 15 - 15.23 The equilibrium constant for the...Ch. 15 - 15.24 Consider the reaction:
If the equilibrium...Ch. 15 - 15.25 A reaction vessel contains at equilibrium...Ch. 15 - 15.26 The equilibrium constant Kc for the...Ch. 15 - At equilibrium, the pressure of the reacting...Ch. 15 - The equilibrium constant K p for the reaction: PCl...Ch. 15 - Ammonium carbamate ( NH 4 CO 2 NH 2 ) decomposes...Ch. 15 - The following equilibrium constants were...Ch. 15 - 15.31 At a certain temperature, the following...Ch. 15 - 15.32 Pure phosgene gas , was placed in a 1.50-L...Ch. 15 - Consider the equilibrium: 2 NOBr( g ) ⇄ 2 NO( g...Ch. 15 - The following equilibrium constants have been...Ch. 15 - 15.35 The following equilibrium constants have...Ch. 15 - 15.36 The equilibrium constant for the reaction at...Ch. 15 - The following diagrams represent the equilibrium...Ch. 15 - 15.38 Outline the steps for calculating the...Ch. 15 - 15.39 The equilibrium constant K? for the...Ch. 15 - 15.40 For the synthesis of ammonia:
the...Ch. 15 - For the reaction: H 2 ( g ) + CO 2 ( g ) ⇄ H 2 O (...Ch. 15 - At 1000 K, a sample of pure NO, gas decomposes:...Ch. 15 - The equilibrium constant K c for the reaction H 2...Ch. 15 - The dissociation of molecular iodine into iodine...Ch. 15 - The equilibrium constant Kc for the decomposition...Ch. 15 - 15.46 Consider the following equilibrium process...Ch. 15 - 15.47 Consider the heterogeneous equilibrium...Ch. 15 - The equilibrium constant K c for the reaction: H 2...Ch. 15 - The aqueous reaction: L-glutamate + pyruvate ⇄...Ch. 15 - 15.50 Explain Le Châtelier’s principle. How does...Ch. 15 - Use Le Chatelier's principle to explain why the...Ch. 15 - 15.52 List four factors that can shift the...Ch. 15 - Does the addition of a catalyst have any effects...Ch. 15 - 15.54 Consider the following equilibrium system...Ch. 15 - 15.55 Heating solid sodium bicarbonate in a closed...Ch. 15 - 15.56 Consider the following equilibrium...Ch. 15 - 15.57 What effect does an increase in pressure...Ch. 15 - Prob. 58QPCh. 15 - Consider the following equilibrium process: PCl 5...Ch. 15 - Consider the reaction: 2SO 2 ( g ) ⇄ 2 SO 3 ( g )...Ch. 15 - In the uncatalyzed reaction: N 2 O 4 ( g ) ⇄ 2 NO...Ch. 15 - 15.62 Consider the gas-phase reaction:
Predict...Ch. 15 - Consider the following equilibrium reaction in a...Ch. 15 - 15.64 The following diagrams show the reaction at...Ch. 15 - 15.65 The following diagrams show an equilibrium...Ch. 15 - 15.66 Consider the reaction . The first diagram...Ch. 15 - Prob. 67APCh. 15 - Consider the equilibrium system 3A → B . Sketch...Ch. 15 - Baking soda (sodium bicarbonate) undergoes thermal...Ch. 15 - Consider the following reaction at equilibrium: A...Ch. 15 - Prob. 71APCh. 15 - 15.72 Consider the following reacting...Ch. 15 - 15.73 At a certain temperature and a total...Ch. 15 - The decomposition of ammonium hydrogen sulfide: N...Ch. 15 - 15.75 Consider the following reaction at a certain...Ch. 15 - When heated, ammonium carbamate decomposes as...Ch. 15 - A mixture of 0 .47 mole of H2 and 3 .59 moles of...Ch. 15 - When heated at high temperatures, iodine vapor...Ch. 15 - 15.79 One mole of and three moles of are placed...Ch. 15 - At 1130°C , the equilibrium constant ( K c ) for...Ch. 15 - For the purpose of determining K p using Equation...Ch. 15 - The following diagram represents a gas-phase...Ch. 15 - 15.83 Consider the following reaction at
When...Ch. 15 - 15.84 A quantity of 0.20 mole of carbon dioxide...Ch. 15 - 15.85 When dissolved in water, glucose (com sugar)...Ch. 15 - 15 86 At room temperature, solid iodine is in...Ch. 15 - 15.87 A student placed a few ice cubes in a...Ch. 15 - 15.88 A mixture containing 3.9 moles of and 0.88...Ch. 15 - 15.89 The equilibrium constant for the...Ch. 15 - When heated, a gaseous compound A dissociates as...Ch. 15 - 15.91 When a gas was heated under atmospheric...Ch. 15 - The first diagram represents a system at...Ch. 15 - A sealed glass bulb contains a mixture of NO 2 and...Ch. 15 - At 20°C , the vapor pressure of water is 0.0231...Ch. 15 - A 2.50-mol sample of NOCl was initially in a...Ch. 15 - 15.96 About 75 percent of hydrogen for industrial...Ch. 15 - Water is a very weak electrolyte that undergoes...Ch. 15 - 15.98 Consider the following reaction, which takes...Ch. 15 - The equilibrium constant Kc for the reaction: 2NH...Ch. 15 - At 25°C, a mixture of NO 2 and N 2 O 4 gases are...Ch. 15 - 15.101 Consider the reaction between and in a...Ch. 15 - In 1899 the German chemist Ludwig Mond developed a...Ch. 15 - For which of the following reactions is K c equal...Ch. 15 - The equilibrium constant K p for the following...Ch. 15 - At 1024°C, , the pressure of oxygen gas from the...Ch. 15 - 15.06 The equilibrium constant for the following...Ch. 15 - 15.107 Industrially, sodium metal is obtained by...Ch. 15 - Consider the equilibrium reaction described in...Ch. 15 - The K p for the reaction: SO 2 Cl 2 ( g ) ⇄ SO 2 (...Ch. 15 - The "boat" form and the “chair" form of...Ch. 15 - A quantity of 6.75 g of SO 2 Cl 2 was placed in a...Ch. 15 - 15.112 Industrial production of ammonia from...Ch. 15 - 15.113 The equilibrium constant for the formation...Ch. 15 - Consider the reaction: 2NO( g )+ O 2 ( g ) ⇄ 2N O...Ch. 15 - The formation of SO 3 from SO 2 and O 2 is an...Ch. 15 - At 25°C , the equilibrium partial pressures of N O...Ch. 15 - 15.117 The vapor pressure of mercury is 0.0020...Ch. 15 - 15.118 Both ' and are important biological ions....Ch. 15 - Photosynthesis can be represented by: 6C O 2 ( g...Ch. 15 - Consider the decomposition of ammonium chloride at...Ch. 15 - 15.121 Eggshells are composed mostly of calcium...Ch. 15 - In the gas phase, nitrogen dioxide is actually a...Ch. 15 - Consider the potential-energy diagrams for two...Ch. 15 - Iodine is sparingly soluble in water but much more...Ch. 15 - The dependence of the equilibrium constant of a...Ch. 15 - Lime ( CaO ) is used to prevent SO 2 from escaping...Ch. 15 - Lime is used to prevent from escaping from the...Ch. 15 - Lime ( CaO ) is used to prevent SO 2 from escaping...Ch. 15 - Lime ( CaO ) is used to prevent SO 2 from escaping...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- At 1130°C, the equilibrium constant (Kc) for the reaction 2H2S(g) ⇌ 2H2(g) + S2(g) is 2.25 × 10−4. If [H2S] = 4.80 × 10−3 M and [H2] = 1.40 × 10−3 M, calculate [S2]. Please write to text formet answerarrow_forwardReaction of nitrogen and hydrogen in the presence of a catalyst produces ammonia: N2 + 3H2 2NH3 , rHo = -92 kJ mol-1 . What would increase equilibrium concentration of the product in the mixture? A) Increase pressure, keep temperature constant B)Increase temperature, keep pressure constant C) Decrease pressure, keep temperature constant D) Information is insufficient for answerarrow_forwardHeating a metal carbonate leads to decomposition. BaCO3(s) BaO(s) + CO2(g) Predict the effect on the equilibrium of each change listed below. Answer by choosing (i) no change, (ii) shifts left or (iii) shifts right. (a) add BaCO3 (b) add CO2 (c) add BaO (d) raise the temperature (e) increase the volume of the flask containing the reactionarrow_forward
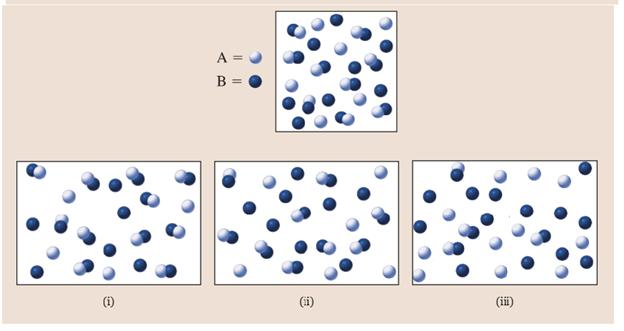
- Suppose you are given the reaction A(g) + B(g) C(g) + D(g). You have the gases A, B, C, and D at equilibrium. Upon adding gas B, the value of K: Select one: a. increases, because by adding A more products are made, increasing the product to reactant ratio b. does not change, as long as the temperature is constant c. depends on whether the reaction is endothermic or exothermic d. does not change, because A does not figure into the product to reactant ratio e. decreases, because A is a reactant so the product to reactant ratio decreasesarrow_forward2C2H2(g) + 5O2(g) 4CO2(g) + 2H2O(g) You have 0.4 moles C2H2 and 0.2 moles O2 present initially in a 5L flask. You allow the reaction to run until it reaches equilibrium. Which of the following is the correct ICE table for this process? Question 5 options: Conc. C2H4 O2 CO2 H2O Initial 0.4 0.2 0 0 Change -x -x +x +x Equil. 0.4-x 0.2-x x x Conc. C2H4 O2 CO2 H2O Initial 0.08 0.04 0 0 Change -2x -5x +4x +2x Equil. 0.08-2x 0.04-5x 4x 2x Conc. C2H4 O2 CO2 H2O Initial 0.4 0.2 0 0 Change -2x -5x +4x +2x Equil. 0.4-2x 0.2-5x 4x 2x Conc. C2H4 O2 CO2 H2O Initial 0.08 0.04 0 0 Change -20x -20x +20x +20x Equil. 0.08-20x 0.04-20x 20x 20xarrow_forwardIf the Kc for this reaction is 9/4, which of the following diagrams represents the molecules in an equilibrium mixture? In the diagrams, A atoms are orange and B atoms are green. Picture Attachedarrow_forward
- Solve all part please. Consider the equilibrium reaction N2O4 (g) ⇌ 2 NO2 (g)a. Using appendix C, find H and S for this reaction (you will need this for b,c). delta s= 176.6J delta H=5802kJb. Knowing that K is temperature dependent and that G = -RTlnK, find the equilibrium constant (K) and T whenthe above reaction is at equilibrium with PNO2 = PN2O4 = 1.0 atm.c. Knowing that K is temperature dependent and that G = -RTlnK, find the equilibrium constant (K) and T whenthe above reaction is at equilibrium with PT = 10.0 atm and PNO2 = 2 PN2O4.arrow_forwardThe reaction shown below has an equilibrium constant value of Kp=5.50 at a certain temperature.CO (g) + Cl₂ (g) ⇄ COCl₂ (g) Kp = 5.50A container is pressurized with CO and Cl₂ until the pressure of CO is 0.450 atm and the pressure of Cl₂ is 0.200 atm. Calculate the pressure of COCl₂ in the container when the system reaches equilibrium.Answer: atm COCl₂arrow_forwardAt a given temperature, a solid sample of PH3BCl3(s) is placed in a closed vessel and allow to reach equilibrium. The reaction is given below: PH3BCl3(s) ⇌ PH3(g) + BCl3(g) Kc = 7.31× 10-3 What is the equilibrium concentration of PH3(g)? Express your answer in M using at least two significant figures. Do not use scientific notation.arrow_forward
- The following combustion reaction is exothermic. If the reaction is at equilibrium, which changes will cause the reaction to shift toward the products? Select all that apply. C2H4(g) + 3 O2(g) ⇌ 2 H2O(g) + 2 CO2(g) a.lowering the temperature b.decreasing the volume c.adding H2O(g) d.removing CO2(g) e.adding C2H4(g)arrow_forwardConsider the following equilibrium: 2HF(g)⇌H2(g)+F2(g) At equilibrium, the concentration of hydrogen fluoride gas, hydrogen gas, and fluorine gas are 2.04 mol/L, 2.47 mol/L, and 1.89 mol/L. What is the value of the equilibrium constant? Answer to 3 decimal places.arrow_forwardCarbon monoxide and hydrogen react according to the following equation: CO (g) + H2 (g) CH4 (g) + H2O (g) When 1.00 mol of CO and 3.00 moles of H2 are placed in a 10.0 L container at 927 ° C (1200 K) and allowed to reach equilibrium, the mixture is found to have 0.363 moles of H2O What is the molar composition (that is, how many moles) of H2 are present in the equilibrium mixture? Answer:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY